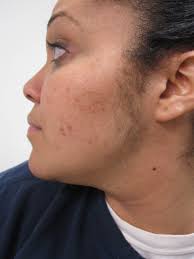
 Typical patient has history of irregular menses, appear hirsute, have acne and in severe cases may be alopecic.
Typical patient has history of irregular menses, appear hirsute, have acne and in severe cases may be alopecic.
PCOS is the most common endocrinopathy among reproductive aged people with ovaries, with an estimated prevalence between six and 13%.
Presence of two or more of the following: chronic oligo-ovulation, or anovulation, androgen excess, and polycystic ovaries.
Polycystic refers to a large number of normal follicles or eggs in the ovary, but does not indicate the presence of large painful cysts.
Affects up to 19.9% of reproductive age women.
Most frequent endocrinopathy in women.
Most common cause of anovulatory infertility in developed countries.
Diagnostic criteria include anovulatin or oligo-avulation, cinical findings or biochemical evidence of hyperandrogenism, ovarian polycystic findings on ultrasound examination, and the absence of other causes for hormonal dysruption.
Rotterdam criteria by ultrasound requires that at least one ovary have 12 or more follicles measuring 2-9 mm in diameter.
The syndrome can be mimicked by congenital adrenal hyperplasia, hyperprolactinemia, acromegaly, Cushing’s syndrome, androgen secreting tumors of the adrenal gland, or ovary, insulin resistance syndromes, androgen use and use of valproic acid.
50% of women with syndrome are obese.
Demonstration of ovulatory dysfunction and hyperandrogenism can be made by hormonal measurements.
Source of excess androgen is mainly from the ovarian theca cells, but some patients have hyperandrogenemia from the adrenal glands
Associated with increased risk of insulin resistance, type II diabetes, dyslipidemia, endometrial carcinoma and psychosocial problems.
Majority of women have a form of insulin resistance, intrinsic to the syndrome, and obese women have added process of insulin resistance related to excess fat.
Anovulatory cycles are common and lead to unopposed estrogen exposure and in turn a lack of cyclic progesterone exposure causes unscheduled episodes of menorrhagia and likely underlies the increased risk of endometrial cancer in PCOS.
Strategy of management should include countering the effects of unopposed estrogen, including the use of combined hormonal contraceptives, cyclic progestins, or progestin containing intrauterine device.
PCOS associated with biomarkers of sub clinical atherosclerosis such as coronary and aortic calcification, greater carotid intima-media thickness, and endothelial dysfunction compared with healthy controls.
Patients with PCOS exhibit endothelial dysfunction about 36% of the time.
Non-alcoholic fatty liver disease more prevalent in PCOS than in healthy controls.
Type 2 diabetes ten times the prevalence as among young normal women.
About 30% of adolescent girls with this syndrome have glucose intolerance or diabetes.
Type 2 diabetes develops by age 30 years in 30-50% of obese women.
Insulin resistance responsible for type 2 diabetes, dyslipidemia and hypertension seen in association with the syndrome.
Insulin stimulates ovarian production of androgen by activating its receptors, indicating that the gland remains sensitive or hypersensitive to it, while muscle and fat tissues are resistant.
Metabolic syndrome prevalence is 2-3 times as high among women with polycystic ovary syndrome as among normal females matched for age and BMI.
20% of women under the age of 20 years with this syndrome have metabolic syndrome.
The risk of fatal myocardial infarction twice as high among women with severe oligomenorrhea versus women with normal menses.
Ultrasound may show multiple small ovarian follicles and support the diagnosis.
Characterized by endothelial dysfunction that is associated with elevated levels of androgen and obesity/insulin resistance.
Most common presentation is anovulatory infertility in 45% of patients.
5-10% of females in the reproductive age group with this disorder have difficulty becoming pregnant.
One in three pregnancies among individuals with PCOS is complicated by gestational, diabetes, preeclampsia, or pre-term birth.
Other pregnancy related risk associated with PCOS include: gestational hypertension, increased risk of neonatal intensive care unit admission.
Anovulation, early pregnancy loss and other pregnancy complications implicated in the low fertility rate.
Most common cause of androgen excess.
Combined hormonal contraceptives lower androgen production and increase sex hormone binding globulin are recommended to treat clinical hyperandrogenism.
Anti-androgen medications such as spironolactone may be added if needed.
For decreased ovulation frequency:
First-line treatment is clomiphene citrate with a resultant ovulatory rate of greater than 80%.
15-20% of patients treated with clomiphene remain anovulatory with pregnancy rates of 33-40% and miscarriage rate of 30-40%.
Second-line treatments include gonadotropins or GnRH.
Metformin is an effective treatment for anovulation in patients with polycystic ovarian syndrome.
Metformin restores the menstrual cycle of 65% of patients with polycystic ovarian syndrome and is associated with a 62% ovulation rate.
Metformin treatment improves ovulatory frequency and can improve both insulin resistance and hyperandrgenism
Metformin treatment in PCOS is associated with weight loss and improved plasma lipid profile.
Metformin improves endothelial function in women with PCOS and endothelial dysfunction independent of changes in glucose metabolism metabolism, dyslipidemia or presence of pre-diabetes.
Metformin has a direct effect on endothelial function in PCOS.
Ovarian wedge resection restores menstrual regularity in 95% of cases with a pregnancy rate of 85%.
Laparoscopic wedge resection results in a 60% pregnancy rate.
Wedge resections result in postoperative adhesions in 36% of cases.
Wedge resections result in loss of significant ovarian tissue and may result in ovarian failure making the procedures obsolete.
Formation of multiple holes on the surface of the ovary by laparoscopy, ovarian drilling, results in menstrual cycle restoration ad ovulation in approximately 80% of women.
Treatment focuses on amelioration of clinical symptoms and restoration of ovulatory cycles so that pregnancy can be accomplished.
Once pregnant patients with PCOS have twofold increased risk of gestational diabetes, preeclampsia, and preterm birth compared to those without PCOS.
Hyperinsulism correlates with increased androgen levels and treatment of insulin excess improves androgenic features of many patients.
Insulin acts synergistically with luteinizing hormone increasing the production of androgen by ovarian theca cells and decreases the sex hormone binding globulin.
Insulin resistance is detectable in the majority of individuals contributing to reproductive metabolic dysfunction and patient should be assessed as to their glucose tolerance, hemoglobin A1c, and they should be screened for cardiovascular disease risk factors including obesity, dyslipidemia, hypertension, and non-alcoholic fatty liver disease.
3-D ultrasound indicates the mean number of follicles per ovary and the mean ovarian volume in a single sonographic plane is greater for women with polycystic ovarian syndrome than for normoandrogenic women.
In women with polycystic ovary syndrome Letrozole outperformed the first-line treatment clomiphene, with higher rates of live births
In a double-blind, randomized, multicenter trial 750 women with POS ages 18 to 40, 27.5% of women treated with 2.5 mg letrozole daily carried a baby to term compared with 19.1% of women treated with 50 mg clomiphene daily (Legro RS et al).
Clomiphene Is the first-line of treatment for infertility in women with polycystic ovary syndrome.
Women treated with clomiphene showed improvements in biochemical hyperandrogenemia, and hirsutism compared to Letrozole.
Women treated with letrozole had a sharper decrease in antral follicle count, a slighter increase in endometrial thickness, and lower estradiol levels during the midluteal phase.
Both clomiphene and letrozole have a risk of serious side effects, and carry an FDA pregnancy category X categorization.
Myo-inositol supplementation during pregnancy for individuals with PCOS does not reduce gestational diabetes, preeclampsia, or preterm birth compared with placebo.
