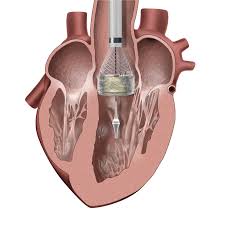
 TAVR uses the transfemoral approach for the treatment of severe, symptomatic aortic stenosis in patients who are inoperable.
TAVR uses the transfemoral approach for the treatment of severe, symptomatic aortic stenosis in patients who are inoperable.
Symptomatic severe aortic stenosis has a two-year mortality rate of nearly 50%.
Uses a TAVR medical device.
Employs an aortic prosthesis either an self-expandable or balloon-expandable system.
Use improves survival as compared with medical therapy in patients with severe aortic stenosis who cannot undergo surgery.
Aortic valve replacement is indicated for patients with severe aortic stenosis with aortic valve area less than 1 cm², who have symptoms, have left ventricle dysfunction, or undergoing cardiac surgery for another indication.
Nearly 30% of patients with severe symptomatic aortic stenosis are not candidates for surgical aortic valve replacement because of multiple medical comorbidities, advanced age, prior surgery, or high risk anatomic features.
Balloon expandable TAVR and surgical aortic valve replacement have similar survival rates at one year among patients considered to be high surgical risk, although the frequency of neurologic events is higher among patients treated with balloon expandable TAVR than among those treated surgically.
Randomized clinical trials involving patients who have less than a 3% risk of surgical operative mortality suggest that TAVR might be the preferred treatment strategy for this particular patient group for aortic stenosis.
With the introduction of TAVR, the number of aortic valve interventions performed in the US has doubled, with a decrease in the annual surgical volume of approximately 45%.
Among individuals older than 80 years of age, the annual number of surgical aortic valve replacement has decreased by 96% because these patients are almost exclusively choosing between TAVR and medical management.
Aortic stenosis and coronary artery disease, share common characteristics, risk factors and clinical presentation.
Coronary artery disease is present in approximately 50% of patient undergoing TAVI.
The use of a renin-angiotensin system inhibitors are associated with lower mortality and risk for heart failure readmission.
Among patients undergoing TAVR device implantation success is achieved in 92% of cases with an overall in-hospital mortality of 5.5%, and a stroke rate of 2% (Mack, MJ et al).
Short term mortality and major complications are relatively low with TAVR with a 30 day mortality of less than 4%, incidence of stroke, less than 2%, and incidence of major bleeding less than 3% with most patients have clinically significant improvement in health status within a month after the procedure.
The rate of 30 day stroke over the first 5 years post TAVR is 2.3%.
Stroke that is related to the transaortic valve implantation (TAVI) can be caused by embolism, hemorrhage, or cardio vascular collapse with cerebral hypoperfusion.
Early periprocedural events of stroke are largely embolic and procedural and origin and arise from manipulation of heavy, calcified aortic valves or the aorta.
Embolization may entail dislodgment of thrombotic, atheromatous, or valve debris during catheter manipulation in valve deployment.
Thrombotic and tissue fragments account for 60 to 70% of emboli, whereas foreign material from catheters and delivery systems accounts for the rest.
Hypotension during rapid pacing or valve deployment can exacerbate the risk of cerebral hypoperfusion.
Late onset strokes tend to reflect coexisting conditions new onset atrial fibrillation, progression of cerebral vascular disease, and other diseases.
MRI imaging steady show that new sub clinical cerebral ischemic lesions are detectable in 68 to 98% of patients.
Presently only 2 valves are approved in the United States: 1, device a balloon expandable (BE) valve made of bovine pericardium mounted in a cylindrical, short stent and 2,self-expanding (SE) valve made of porcine pericardium mounted in the tall or, nitinol stent with an adaptive shape.
The BE valve is also approved for patients with aortic stenosis or operable but are at high risk for surgery.
Studies of the above valves have shown 30 day outcomes ranging from 5%-9.6% for mortality, 1.5%-4.3% for stroke, 0.3-1.2% for myocardial infarction, 6.7%-21.5% for moderate paravalvular regurgitation and 6-24.2% for post CABG or permanent pacemaker placement (Blackman DJ et al, DiMario C et al, Chieffo A et al).
In a clinical trial comparing BE and SE for device success the BE valve implantation resulted in a higher device excess than the SE, 95.9% versus 77.5%, respectively (Abdel-Wahab et al).
In the above study there was no difference in 30 day cardiovascular mortality rates: 4.3% in the SE valve group and 4.1% in the BE valve group.
Both valves have good flow characteristics.
SE valve use associated with high rate of residual paravalvular aortic regurgitation.
Surgical AVR has less paravalvular regurgitation rates then TAVR.
30 day mortality rates are higher and more variable in hospitals with low procedural volume than in hospitals with a high procedural volume.
Thromboembolic complications such as stroke, systemic embolism, valve thrombosis, and venous thromboembolism have been described after TAVR.
Presently, guidelines recommend the use of dual antiplatelet therapy early after TAVR.
Practice guidelines recommend clopidogrel in addition to aspirin for the first 3-6 months after TAVR in patients who do not have an indication for oral anticoagulation.
The procedure is complicated by life-threatening bleeding in 3-13% of patients and strokes occur in 1-12% at one year after TAVR.
In a study of rivaroxaban versus antiplatelet therapy there is a higher risk of death and thromboembolic complications and a higher risk of bleeding.
Among patients with severe aortic stenosis who were at low surgical risk, the rate of death, stroke, or rehospitalization at one year was significantly lower with TAVR than with surgery (Mack MJ).
Guidelines, recommend transfemoral aortic valve implantation for patients with symptomatic AS who are unable to undergo surgery or have a high estimated surgical mortality risk of greater than 8%.
Guidelines recommend surgical aortic valve replacement for adults younger than 65 years, with a mechanical valve preferred over a bioprosthetic valve in those younger than 50 years based on expected years of remaining life and valve durability.
TAVI is recommended for individuals age 75-80 years and older.
Cerebral embolic protection (CEP) devices are designed to prevent debris release during the TAVR procedure from reaching the brain, by reducing the risk of stroke: it’s routine use however it does not decrease the incidence of stroke within 72 hours.(BHF PROTECT TAVI investigators).
