 Primitive gut begins to form in embryological development as endoderm becomes the intestinal epithelium and glands.
Primitive gut begins to form in embryological development as endoderm becomes the intestinal epithelium and glands.
The health and performance of the gastrointestinal tract is influenced by the interaction of a variety of factors, including diet, nutritional status, genetics, environment, stress, the intestinal microbiota, immune status, and gut barrier.
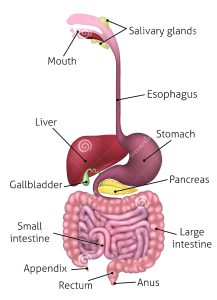
The gastrointestinal tract and associated dorsal mesentery are subdivided into foregut, midgut, and hindgut regions based on the respective blood supply.
The foregut is supplied by the celiac trunk, the midgut is supplied by the superior mesenteric artery, and the hindgut is supplied by the inferior mesenteric artery.
The mesoderm becomes the connective tissue, muscle and intestinal wall beginning during the fourth week of embryological development.
During the 5th week of fetal development the intestine elongates and the midgut loop herniates through the umbilicus, with the cranial limb becomes the distal duodenum to the proximal ileum and the caudal limb becomes the distal ileum to the proximal two-thirds of the transverse colon.
By the 10th week of fetal development the midgut loop returns to the abdominal cavity and rotates 270 degrees counterclockwise around the axis of the superior mesenteric artery and becomes fixed at this site.
The main purpose of the gastrointestinal tract is to digest and absorb nutrients: fat, carbohydrate, protein, micronutrients (vitamins and trace minerals), water, and electrolytes.
Digestion involves both mechanical and enzymatic breakdown of food.
Mechanical processes include chewing, gastric churning, and the mixing in the small intestine.
Enzymatic hydrolysis is initiated by intraluminal processes requiring gastric, pancreatic, and biliary secretions.
Gastrointestinal tract secretions:
Saliva 500-1500 cc/d
Stomach 1500-2500 cc/d
Bile 500-1000 cc/d
Pancreas 600-1500cc/d
Intestine 1000-1800cc/d
This 7-8 L load of upper GI tract secretions is reduced by the small and large intestines to less than 200 g of stool that contains less than 8 g of fat, 1–2 g of nitrogen, and less than 20 mmol each of Na+, K+, Cl–, HCO3–, Ca2+, or Mg2+.
Gastric secretions are rich in hydrochloric acid.
The secretion of hydrochloric acid by the stomach stimulates bicarbonate secretion by the pancreas once the HCl reaches the duodenum.
The entire lining of the inside is replaced approximately every seven days.
Pancreatic secretions neutralize the gastric secretions.
Pancreatic secretions responsible for most of the digestion of triglycerides in the upper small intestine.
Lipase present in high concentration in pancreatic secretions and its catalytic activity digests dietary fat.
Fluid from the body enters the intestinal lumen during digestion.
This fluid is isosmotic with the blood and contains a high quantity, about 142 mEq/L, of sodium.
A healthy individual secretes 2000–3000 milligrams of sodium per day into the intestinal lumen.
Bile salts have an important role in the lipolysis of triglycerides by pancreatic lipase as well as in the transportation of digested and fat soluble vitamins across the the intestinal mucosa.
Saturated fats and fat soluble vitamins are dependent on micellar solubilization for absorption (Barrett KE).
Bile salts and pancreatic enzymes combine to absorb 97% of ingested fat in the first 5 feet of the small intestine.
Bile salt pool is recycled twice per meal and up to 6-8 times a day.
95% bile salts are recycled via the enterohepatic circulation within the last 100 cm of ileum.
Loss of greater than 100 cm of ileum creates a bile salt deficiency.
Bile salt deficiency, can decrease fat absorption by up to 40-50%.
Bile deficiency or insufficiency causes include: primary biliary cirrhosis, primary sclerosing cholangitis, cirrhosis, cholestatic processes, external bile drainage, gastric hypersecretion, distal bile duct obstruction, bacterial small bowel overgrowth, impaired enterohepatic obstruction with ileum resection or fistula disease (Parrish CR).
The normal intestinal epithelium is comprised of a single layer of epithelial cells linked by tight junctions and intercalated with immune cells.
The intestinal epithelium is a dynamic tissue containing a series of villi and crypts.
The intestine functions include nutrient absorption, acting as a physical barrier against gut luminal contents, and responding to signals from the intestinal microbiota and immune system.
The digestive tract the most susceptible part of the body to inflammation and disease: most of our immune cells and the microbes that regulate those immune responses live in our digestive tract.
Secretory cells of the intestinal tract include goblet cells, which produce mucus and such antimicrobial peptides as trefoil factor and resistin-like molecule beta that limit luminal microbes.
Lifetime risk of cancers in the GI tract varies by a factor 24-0.2% small intestine and 4.82% for the large intestine.
Motility of the G.I. tract is regulated by coordinated neurohormonal mechanisms that affecr smooth muscle contractility.
The gut plays several important roles including acting as the main pathway for fluid, electrolyte and nutrient absorption while also acting as a barrier to toxic agents present in the gut lumen including acid, digestive enzymes and gut bacteria.
It is also a major immunological defense mechanism, detecting natural commensals and triggering immune response when toxic microbes are present.
Failure of homeostasis due to trauma, drugs and infectious microbes not only damages the gut but can lead to influx of damaging agents into the bloodstream.
Gut motor activity is primarily controlled by the intrinsic or enteric nervous system.
The CNS modulates gut motor activity the extrinsic sympathetic and parasympathetic pathways, whereas descending pathways in the spinal cord modulate transmission of sensory input from the dorsal horn to supraspinal centers.
Visceral bowel sensation is conveyed by afferents that travel to the spinal cord and ultimately to the cerebral cortex, as well as do the vagus nerve to the brain stem.
There is a 10 to 1 ratio of afferent to efferent fibers in the vagus nerve at the level of the diaphragm.
The vagus nerve primarily conveys subnoxious messages,whereas the spinal afferents convey nonnoxious and noxious input to the dorsal horn of the spinal cord.
Subsequently information is conveyed through the spinothalamic tract to the medial and posterior thalamus subsequently to the primary somatosensory cortex, which localized and discriminates somatic and visceral sensations.
Descending inhibitory fibers from the interior cingulate cortex to the distal horn of the spinal cord use endogenous opioids, serotonin and norepinephrine to modulate or gate viscreal sensation including pain when search messages from the viscea conveued to higher centers.
Functional disorders of the G.I. tract result from the effects of biological, psychological, and social factors.
Luminal gastrointestinal irritants primarily include bile acids, nonabsorbed complex carbohydrates and products of their material bacterial metabolism such a short chain fatty acids.
Bile acids and short chain fatty acids activate secretion of enteroendocrine celll products such as five hydroxytryptamine and stimulate colonic motility and defecation.
Healthy individuals have little awareness of G.I. physiological processes except for the sensation of postprandial fulness after a meal or the desire to defecate.
Individuals may have increased sensitivity due to increased sensitization of peripheral afferent receptors or spinal dorsal horn neurons, alterations in descending modulation, or central amplification.
Sensitization of peripheral receptors may be due to low grade information, which activates nociceptors and increases sensitivity.
