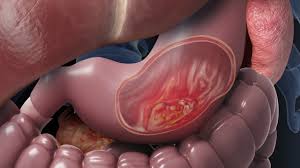
 Stomach cancer is the 5th most common malignant disorder in the world.
Stomach cancer is the 5th most common malignant disorder in the world.
There were more than 968,000 new cases globally, resulting in 660,000 debts in 2022.
In 2020 there was an estimated 768,793 deaths from stomach cancer, which is one of every 13 cancer related deaths.
In 2025 an estimated 30,300 people are to be diagnosed in the US and 10,700 people are expected to die of gastric cancer.
Fifth leading cause of cancer mortality worldwide, with a five-year survival of only 35% among all stages.
There is a marked geographic variation with a 15 to 20 fold difference between high and low incidence regions.
The highest gastric cancer incident rates occur in Northeast Asia, South and Central America and Eastern Europe.
Rates are particularly high in Japan and Korea, where gastric cancer is the most commonly diagnosed cancer in males and China were gastric cancer is the leading cause of cancer related mortality.
In the US gastric cancer is the 15th most common diagnosed cancer and the 15th bleeding cause of cancer related death.
Most patients are diagnosed with advanced disease, making surgical resection either non-feasible or available with purely palliative intent.
Second most common cause of cancer related deaths in men and the fourth most common in women (Kamangar F et al).
Gastric carcinoma incidence is decreasing.
Although gastric cancers are declining in some areas, the number of deaths may increase in the coming decades due to aging populations in high incidence areas of Latin America and Eastern Asia.
Ratio of men to women is about 2:1.
Include adenocarcinomas, carcinoids, neuroendocrine carcinomas and malignant gastrointestinal stromal tumors.
Adenocarcinomas are epithelial derived cancers and are the most common malignancies of the stomach (95%).
The incidence of gastric cancer in the United States is 1/10 that of South Korea.
60% of new cases occur in eastern Asia.
100,000 new cases each year in Japan.
In the US, the incidence is highest in nonHispanic blacks, individuals of Asian and Pacific island descent, and Hispanic and Latino individuals.
In these in populations the incidence is nearly twice that of non-Hispanic white individuals, who have the lowest incidence.
Native American and Alaskan populations have any incidence higher than that of non-Hispanic whites, but lower than that of other ethnic populations.
Among non-Hispanic whites, the rates of death related to gastric cancer occur at approximately half the rate observed in other ethnic populations.
Present staging recognize carcinomas as tumors arising in the distal stomach or those originating in the proximal 5 cm of the stomach but not crossing the gastroesophageal junction.
They are typically classified based on anatomical location cardia/proximal or noncardia/distal and histologic type diffuse or intestinal.
The diffuse type is characterized by poorly differentiated and discohesive tumor cells with signet ring or nonsignet ring morphology diffusely infiltrating the gastric wall in a desmoplastic stroma.
The diffuse type is more prevalent in low risk areas and is mostly associated with heritable genetic abnormalities.
The intestinal type tends the form of mass lesion with cells arranged in tubular glandular pattern with scattered goblet cells and occurs more frequently in high risk areas and accounts for most of the geographic variation seeing with this disease.
The intestinal type gastric cancer is often related to environmental factors such as H pylori infection, tobacco use, high salt intake and other dietary factors, including alcohol.
There is been a more decline in intestinal type gastric cancer of the distal stomach in North America over the past several decades, mainly due to enhanced access to clean drinking water, improved food preservation, a diet with low promotion of gastric cancer, and H. pylori eradication.
Screening for H. pylori antigen does not lower the incident of gastric carcinoma.
The incidence rate of diffuse gastric cancer of the proximal stomach is rising.
Noncardia cancer increasing in young Caucasians under 50 years, especially in women.
Clinical staging is based on results of physical exams, imaging tests and biopsies.
Newly diagnosed patients receive physical examination, CBC, chemistry profile, esophagogastroduodenoscopy with biopsy of the primary lesion, CT scan, enhanced of the chest, abdomen and pelvis, FDG/PET-CT evaluation is recommended for locally advanced or etastatic disease, endoscopic ultrasound is recommended for early stage disease, testing for microsatellite instability status is recommended, HER2 programmed death ligand-1, and Claudin 18 iso form 2 testing is recommended.
Diagnostic laparoscopic surgery is indicated to assess regional and peritoneal disease prior to resection.
Staging is based on size and location of the primary tumor, whether it is spread locally or invaded the adjacent structures , If local lymph nodes are involved, or the tumor has spread distally.
Outcomes for gastroesophgeal tumors after resection differ from outcomes for gastric cancer.
In a phase II international trial ToGA of about 3800 patients with advanced gastric or gastroesophageal cancer, most of the patients from Asia or Europe, were found to have HER-2/neu positive disease in 22% of patients, and the median survival rate for patients receiving Herceptin was 13.8 months compared to 11.1 months in the group that received chemotherapy alone.
Median survival for a patient with stage IV, HER 2-negative gastric cancer is about one year, but for HER 2-positive patient it is about 18 months.
HER2 expression is more common in proximal cancers.
HER2 expressionPositivity in patients with gastric cancer ranges from 12 to 23%.
HER2 expression varies with the histologic subtype intestinal greater than diffuse, and tumor grade, moderately differentiated greater than poorly differentiated.
HER2 positivity in gastric cancer is significantly higher in males versus females, E-G junction tumors versus stomach tumors, and intestinal subtypes versus diffuse subtypes.
HER2 testing is recommended no patients with gastric cancer at diagnosis of metastatic disease as documented was suspected.
Immunohistochemistry and or molecular testing for HER2 status MSI or MMR status, PDL1 expression, tumor mutation burden status, and neutrophilic tropomyosin related kinase gene fusions are implicated in the clinical management of advanced gastric cancer.
MSI or MMR should be performed for all newly diagnosed gastric carcinomas.
Liquid biopsy/next generation sequence studies may help identify tumor treatment targets.
Annual incidence of 5-15 deaths per 100,000 patients in Europe.
Highest rates in Japan, eastern Asia, with high rates in eastern Europe and areas of Latin America.
Most cancers occur in developing countries with estimated 42% of worldwide cases occurring in China.
Unhealthy lifestyles include: tobacco smoke, alcohol consumption, consumption of preserved foods of foods high in salt, and low intake of fresh vegetables or fruits.
Incidence low in western Europe and North America.
Decline in incidence in most economically developed countries due to improved food storage, and lifestyle changes.
Steady decline in gastric cancer’s incidence and mortality is due to reduction in chronic infection with H pylori and increased availability of fresh fruits and vegetables and decreased use of salt preserved food.
There has been a marked decline in intestinal type gastric cancer of the distal stomach over the past several decades, mainly due to enhanced access to clean drinking water, improved food preservation, and average diet with low promotion of gastric cancer, and H pylori eradication.
The incidence of diffuse type gastric cancer of the proximal stomach are rising, and its cause remains elusive.
Tumors of the distal stomach, predominate in low and middle income countries.
H. pylori is less prevalent in wealthy western societies and may account for some of the reduction in the incidence of gastric cancer.
H. pylori. main driver of non-cardia gastric cancer.
H. pylori infection modifies the risk of gastric cancer associated with germline pathogenic variants in homologous recombination genes.
In 1994 6.3 deaths per 100,000 men and 4.2 deaths per 100,000 women in the U.S.
In most countries incidence and deaths twice as high in men than women.
Japan only country with a moderately good survival rate as a result of mass screening.
5 year survival rates are approximately 30%,but in Japan 5 year survival rates are approximately 60%.
5 year survival for gastric cancer entirely confined to the stomach is 67%.
The five-year survival for gastric cancer diagnosed at a regional stage into ajacent tissues, or involves regional lymph nodes, or both, or at a distant stage, the five-year survival rate drops 31% and 5%, respectively.
In Japan it is the most common cancer among men.
In China more new cases of diagnosed each year then any other country.
Malignancies other than adenocarcinomas account for ≤10% of gastric cancers and include: adenocanthomas, adenosquamous, squamous cell carcinoma, parietal cell cancer, oncocytic cancer, carcinoid and gastrointestinal stromal tumor, choriocarcinomas, teratomas, yolk sac tumors, lymphomas and MALToma.
There is greater than 5-fold reductions were found in four miRNAs in tumor-adjacent tissues and gastric cancers as compared to those miRNA levels in normal gastric tissues.
Early gastric cancer is defined as a lesion confined the mucosa regardless of the presence of lymph node metastases.
Metastases to lymph nodes seen in 3-5% of patients with mucosal carcinomas of the stomach.
Proximal tumors associated with obesity and reflux esophagitis.
Proximal and distal lesions have a better prognosis in Asians.
In the Far East most gastric cancers are limited to the gastric mucosa and submucosa with rare spread and have a 10-year survival rate of 80-95%.
In the Western Hemisphere the most common sites of gastric cancer of the proximal lesser curve, cardia and gastroesophageal junction.
There is a marked geographic variation noncardia cancers with high incidence of such lesions in Japan, Korea, China, Taiwan, Costa Rica, Peru, Brazil, Chile, and the former Soviet Union.
Noncardia gastric cancers is likely initiated by chronic inflammation such as by Helicobacter pylori, chronic gastritis or autoimmune gastritis and progresses from chronic gastritis, intestinal metaplasia, and dysplasia.
Noncardia gastric cancers associated with an increased risk from tobacco, high salt intake and alcohol consumption.
Gastric cardia, or upper stomach cancer, is associated with a higher risk due to obesity.
For every kilogram increase in BMI there is a 4% increased risk of developing gastric cardia cancer.
A dose-dependent relationship between smoking and non-cardia gastric cancer risk for distal gastric cancers ranges from 1.5-2.1.
The use of nonsteroidal anti-inflammatory drugs reduces the risk of non-cardia gastric cancers as those eating a diet high in fruits and vegetables.
People who take proton pump inhibitors (PPIs), have more than twice (2.44) the risk of developing stomach cancer, whereas H2 blockers were not associated with an increased risk.
Small percentage of cases related to hereditary factors with majority related to environmental, socioeconomic and dietary factors.
Increased incidence in lower socioeconomic groups and low education level.
Associated with blood group A, ionizing radiation, gastric ulcer and individuals with prior gastric resection.
Higher incidence in individuals who work in coal, nickel, and asbestos mines and in processing of timber and rubber.
Other factors include obesity, heavy drinking, and smoking.
Cigarette smoking estimated to contribute 20% of gastric cancer deaths.
Increasing incidence in proximal tumors.
Prognosis is worse for proximal tumors of the cardia compared to distal gastric lesions.
Gastric cancer generally carries a poor prognosis, as it is often diagnosed in advanced stage.
In Japan and South Korea screening is performed widely, and early detection improves outcomes.
Distal gastric tumors are more common in Asia and have a 5 year overall survival of up to 60%, compared with gastric cardia tumors more common in the US with a 5 year overall survival rate of approximately 20%.
Tumors associated with GERD also more common in obese men who drink alcohol and smoke.
Asian Americans have less metastatic disease and more distal lesions with better overall median survival, and whites and Latinos have similar risk of death.
Risk of death higher for African Americans.
The US incidence rates are 2 to 3 times higher in African-American, Asian and Hispanic populations as compared with White populations.
Hereditary gastric cancer associated with an autosomal dominant pattern to develop diffuse gastric cancer which are poorly differentiated adenocarcinomas infiltrating a thickened stomach wall without a specific mass.
Hereditary diffuse gastric cancer occur before age 40 years with an average onset of 38 years, associated with germline mutations in E-cathedrin and CDH1 genes.
Hereditary diffuse gastric cancer defined associated the presence of two or more documented cases of diffuse gastric cancer in 1st or 2nd degree relatives, with at least one case diagnosed before the age 50 or three or more documented cases in 1st or 2nd degree relatives, regardless of age of onset.
Cumulative risk for gastric cancer in CDH1 mutation carriers 70-80% by 80 years for men and 56% for women.
Approximately 40% of hereditary diffuse gastric carcinoma (HDGC) families have a germline mutation in CDH1.
Patients with CDH1 carriers are considered for prophylactic gastrectomy.
Patients with germline pathogenic variants of BRCA1 and BRCA2 in the homologous recombination genes substantially increase the risk of gastric cancer.
Mismatch repair gene abnormalities are associated with increased risk of gastric cancer.
Habitual male smokers are more likely to have well differentiated cancers associated with a longer survival time compared to those with poorly differentiated lesions.
Nonproximal tumors predominate in Japan and other parts of the world.
Helicobacter pylori-induced gastritis strongest risk factor.
Only a small proportion of patients, (less than 5%) with Helicobacter pylori induced gastritis develop gastric cancer during their lifetime.
H. pylori infects more than half of the worlds adult population and chronic infection is a dominant risk factor for gastric cancer, accounting for an estimate two thirds of all the cases globally.
Eradication of H, pylori using amoxicillin and omperazole reduced gastric cancer incidence by 39% after 15 years of follow-up (Ma JL et al).).
H.pylori infection increases the risk of gastric cancer associated with germline pathogenic variants in homologous recombination genes.
Epstein-Barr virus infection is associated with approximately 7.5% of gastric cancers.
Histology of adenocarcinomas divided into intestinal and diffuse types.
Intestinal type of gastric cancer seen in countries with high incidence rates and referred to as endemic form of cancer.
Intestinal type of adenocarcinoma progresses throug a sequence of lseions induced by H. pylori such as chronic gastritis, rophic gastritis, intestinal metaplasia, dysplasia, and adenocarcinoma.
Intestinal type gastric cancer has well defined ductal structures with desmoplstic stroma reaction with inflammatory infiltration.
Intestinal type arises in the antrum or antral-corpus junction.
Diffuse type lesions involve the corpus of the stomach.
Diffuse types of gastric adenocarcinoma consistent of individually infiltrating malignant cells that did not form glandular structures.
Diffuse type lesions have no known precursor lesions, although mutations or silencing of E-cadherin gene may be associated carcinogenic events.
The E-cadherin (CDH1) gene, which plays an intricate role in diffuse gastric cancer.
E-cadherin is a cell adhesion molecule that is encoded on chromosome 16, and is responsible for cell-to-cell adhesion between epithelial cells, forming a barrier between adjacent cells.
When E-cadherin is mutated, it allows cancer cells to invade previously impenetrable borders and can contribute to metastases.
The loss of E-cadherins has been implicated in gastric, colorectal, lobular breast, and ovarian cancers.
With a mutated CDH1 gene leading to an E-cadherin deficiency there is a greater than 80% chance of acquiring diffuse gastric cancer in their lifetime.
Patients with CD1 gene typically present around age 40 years.
Majority of patients with hereditary diffuse cancer syndrome do not have a germline mutation in the one known causative gene R-Cadherin (CDH1).
A gastric cancer study in twins estimated the heritability of gastric cancer is approximately 22%.
Patients with this known mutation are usually advised to have a total gastrectomy sometime between 20 and 40 years of age.
Diffuse gastric cancer is difficult to diagnose as tumor cells are typically located below an intact mucosa and can be easily missed, even with extensive biopsies.
Diffuse gastric carcinoma often referred to as linitis plastica due to its appearance on inspection that resembles a leather bottle.
Diffuse gastric cancer associated with unintended weight loss, nausea and vomiting, early satiety, and may have a strong family history of cancer, especially involving gastric, colorectal, ovarian, and breast cancer.
Intestinal type of gastric cancer is responsible for global ethnic variation while he incidence of diffuse is approximately the same regardless of geography or race (Munoz N).
Diffuse gastric cancer and occurs at an incidence of 4.7 per 100,000 people compared to 8.6 100,000 people for intestinal gastric cancer (Ekstrom AM).
Endoscopic ultrasound helps to determine the T classification and can assess the presence of regional lymph node metastases.
Endo scopic ultrasound accurately discriminated T1/T2 tumor from T3/T4.
Many institutions employ staging laparoscopic evaluation in patients with potentially resectable gastric cancer.
Blummer’s shelf-rectal mass related to metastatic gastric cancer.
Virchow’s node=supraclavicular lymph node.
Decrease in gastric cancer has occurred in the endemic or intestinal form. This form is seen in high incidence countries and results from a combination of achlorhydria, migration of small intestinal epithelium and superinfection with Helicobacter pylori.
Diffuse cancers may be associated with H. pylori but generally not associated with intestinal metaplasia.
Diffuse type/signet cell histology correlates with poorer outcomes, and a predilection for intraperitoneal metastases when compared to intestinal type lesions.
1-3% of such lesions associated with inherited predisposition syndromes.
E-cadherin ,mutations occur in approximately 25% of families with autosomal dominant predisposition to diffuse type gastric cancer.
Anti-actin antibodies may be present and they are associated with a poor prognosis.
Prophylactic gastrectomy should be considered in young patients with CDH1 mutation and family history of hereditary diffuse gastric cancer.
Criteria for CDH1 molecular genetic testing include: 2 or more cases of gastric cancer, with at least one diffuse gastric cancer diagnosed before age 50, three or more cases of gastric cancer in a family diagnosed at any age, with at least one case of diffuse gastric cancer, an individual with a diffuse gastric cancer before age 45 years, a case of both diffuse gastric cancer and lobular cancer of the breast, and one family member with diffuse gastric cancer and another member with signet cell cancer of the colon.
Approximately 1 to 3% of gastric cancers are associated with an identified genetic predisposition.
Germline variant and genes such as CDH1, CTNNA1, or PALB2 are associated with increased to hereditary diffuse gastric cancer.
Other hereditary syndromes include Lynch syndrome-MLH one, MSH2, MSH6, and PMS2, juvenile polyposis syndrome, Puetz Jeghers syndrome, Familial adenomatous polyposis, and hereditary breast and ovarian cancer syndrome/BRCA1 and BRCA2.
Among patients with H. pylori infection who had a family history of gastric cancer in first-degree relatives, H. Pylori eradication treatment reduced the risk of gastric cancer.
Management options for CDH1 carriers include prophylactic gastrectomy or intense surveillance for early detection and treatment.
Evaluation of newly diagnosed patients include a complete history and physical exam, CBC, chemistry profile, upper G.I. endoscopy with biopsy of the primary tumor, CT scan with contrast of the chest, abdomen and pelvis, and PET/CT may also be recommended.
Endoscopic resection maybe therapeutic for early stage disease.
Universal testing for MSI or MMR is recommended in all newly diagnosed patients.
HER2 and PD-L1 testing is recommended that the time of diagnosis if metastatic disease is present or suspected.
Initial staging classifies patients into three clinical groups localized cancer stages cTis or cT1a, local regional cancer cancer stages cT1b-cT4a ; cM0, and metastatic cancer stage cT4b; cM1
U.S. National Cancer Data Base reporting on 50,169 cases treated with gastrectomy indicated an overall survival rate of 63% at 1 year and a 28% survival at 5 years.
Management of unresectable or metastatic disease may include systemic therapy and or chemo radiation to provide symptom relief and delay progression.
Systemic therapy compared to palliative supportive care for advanced gastric cancer is associated with a survivor benefit: with overall survival eight months versus five months.
Surveillance, Epidemiology, and End Results (SEER) Data demonstrates overall 5 year survival of approximately 30% due to most patients presenting with locally advanced disease.
5 year survival for patients with localized disease at diagnosis is 62.3%, and 27.7% in patients with lymph node positive disease, 3.7% patients with metastatic disease.
Endoscopic ultrasound helps to determine the T. classification and can assess the presence of regional lymph node metastases.
Many institutions employ staging laparoscopic evaluation in patients with potentially resectable gastric cancer.
Appropriate staging requires the histological evaluation of at least 15 lymph nodes at the time of gastric resection.
T-1 lesions involving the sub mucosa associated with lymph node involvement in about 19% of cases.
T1 a or b cancers could be treated the endoscopically or by limited surgery including the laparoscopic approach, but higher stages require more involved therapies.
For T2 lesions, which invade the muscularis propria or submucosa, lymph node metastases are seen and greater than 50% of cases.
Evidence exists that some patients with lymph node metastases beyond the perigastric lymph nodes can be cured with surgical resection alone (Gotoda T).
D1 lymphadenectomy encompasses all anatomically defined N1 node stations for a given location of tumor.
D2 lymphadenectomy encompasses all D2 node stations.
D1 resection refers to removal of regional perigastric lymph nodes was compared with D2 resection which refers to extended lymphadenectomy to include level 1 and 2 regional nodes.
Radical gastrectomy with extended, D2, removal of regional lymph nodes is standard treatment for curable cancer in Japan.
D2 resection-extended lymph node dissection, entailing removal of nodes along the hepatic, left gastric, celiac, and splenic arteries, as well as those of the splenic hilum.
Partial or complete gastric resection with lymphadenectomy is the only potentially curative therapy for early gastric cancer, and the use of perioperative or adjuvant chemotherapy improves overall survival.
Despite multi modality therapies more than 60% of patients after gastric resection develop locally recurrent or metastatic disease.
Adjuvant therapy improves cure rate by at least 10% of what can be achieved by surgery alone.
Adjuvant or neoadjuvant chemo therapy is the standard of care for medically fit patients with stage II- III gastric cancer.
80-90% of patients in Western countries are diagnosed at an advanced stage, with unresectable disease, or have disease relapse within 5 years after initial curative intent surgery.
In the US 67% of patients present with stage II or IV disease and only ten cent with stage I disease.
Randomized controlled studies in Europe compared D1 with D2 lymphadenectomy and failed to show a benefit for the D2 resection, but lack of experience with the surgical procedure was felt to account for the poor outcome in D2 cases.
American Intergroup 0116 study of chemoradiation after limited resection of lymph nodes, D0 or D1, decreased the local recurrence rate and increased long term survival, indicating chemoradiation eliminates residual nodal metastases that could be removed by D2 lymphadenectomy.
D3 lymphadenectomy encompasses all D3 node stations.
Only one third of all patients with newly diagnosed gastric carcinoma can undergo potentially curable surgery.
Surgical management will gastric carcinoma may involve endoscopic mucosal resection for early stage disease, minimally invasive laparoscopic resection, or open gastrectomy for more advanced disease.
Minimally invasive surgery data suggested in randomized studies equivalency compared to open techniques.
Total gastrectomy is used for proximal or middle third lesions, and a partial gastrectomy is recommended for lesions in the distal third of the stomach.
Gastric resection goal is to obtain negative margins (R0) resection, since a positive margin (R1) dissection is associated with a worse prognosis.
Achievement of R0 resection is central for long-term survival.
R0-no cancer at resection margin.
R1-microscopic residual cancer.
R2-macroscopic residual cancer M1.
Overall survival after the gastrectomy is correlated with the total number of lymph nodes removed and even then number of non-malignant lymph nodes.
For every 10 extra lymph nodes dissected, the overall survival was improved by 7.6% after gastrectomy.
Wide resection margin goals are 4-6 cm for potentially curative surgery, because there is propensity for mucosal spread, so that close gross negative margins are not sufficient.
Endoscopic mucosal resection applicable for early lesions, T1, can be utilized in selected cases.
Only 10-20% of patients that present with localized, distal cancer are surgically curable.
Most patients present with advanced or metastatic disease.
In individuals who have undergone gastric resections with curative intent, the recurrence rate approximates 50%.
Autopsy studies examining patients after gastric resection found 80-93% of patients had local regional relapse, and 49% demonstrated distant relapse (Dikken JL et al), suggesting the necessity of adjuvant chemotherapy.
Meta-analyses of 17 worldwide randomized trials of postoperative chemotherapy versus surgery alone demonstrates a significant improvement in overall survival and disease-free survival, as well as significant improvement in median survival.
In east Asia, particularly in Japan ad South Korea, D2 gastrectomy is the standard surgical treatment for localised gastric cancer.
D2 resection recommended for resectable disease.
In the CLASSIC trial, a phase 3 trial of adjuvant chemotherapy in Asian patients after gastric surgery, 1035 patient’s stage II-IIIb were assigned to adjuvant chemotherapy with capecitabine and oxaliplatin for 6 months with surgery alone: Disease-free interval was improved across all stages (Bang-YJ et al).
Most patients will require systemic therapy during the course of their illness.
Distal cancers associated with low socioeconomic status, high salt intake, Helicobacter pylori infection and chronic atrophic gastritis.
Risk factors include adenomatous gastric polyps, prior gastric surgery with a 20 year latency period and pernicious anemia.
Additional fat risk factors include family history of gastric cancer, tobacco smoking, reported salty taste preference, obesity, and gastroesophageal reflux disease
Associated nutritional factors diet high in nitrates, salted, smoked mets, low intake of vitamins A,C and fruits and vegetables.
The major prognostic factors for patients with gastric cancer are the depth of the primary tumor and the presence or absence of lymph node, peritoneal and distant metastases.
The presence of absence of lymph nodes metastases is the most important predictor of survival following a curative resection.
Palpable lymph nodes in the left supraclavicular space (Virchow node) or left axilla (Irish node) implies incurability.
Peritoneal disease is considered end-Stage disease and the combination of cytoreductive surgery and hyperthermic intraperitoneal chemotherapy is a consideration: can improve disease free and overall survival.
Periumbilical nodules (Sister Mary Joseph Node) indicates the presence of peritoneal dissemination of malignancy.
Krukenberg tumors, ovarian metastases, or peritoneal metastases (Blumer shelf) reflect drop metastases.
The presence of immunohistologically detected micrometastases in the regional lymph nodes does not affect survival of Japanese patients with pT2N0M0 gastric cancer.
Survival of patients with node positivity after resection is about 30%.
Median survival for metastatic or unresectable disease 7-10 months.
2-year survival with conventional chemotherapy for patients with metastases is 10-15%.
Median survival with combination chemotherapy remains less than 1 year.
Five-year survival approximately 3% for patients diagnosed with metastatic disease.
Response rates for combination of irinotecan and cisplatin range in metastatic disease ranges from 42-58% with a median time to progression from 4.2-5.8 months and median survival time of approximately 9 months.
5-year survival rate less than 20%.
5-year survival rate for patients who undergo curative resection ranges from 47-60%.
Lymph nodes involved in more than 50% of patients at the time of diagnosis.
Diagnosis often delayed for lack of symptoms in early stage disease.
Symptoms frequently vague with weight loss, epigastric discomfort, nausea, vomiting, loss of appetite, fatigue and early satiety.
Most common test for diagnosis is upper endoscopy which provides information about tumor location, extent of involvement, and permits biopsy.
Endoscopic ultrasound can provide information about the depth of tumor penetration into the wall of the stomach and helps in the assessment of lymph node enlargement.
Endoscopic ultrasound can assess the depth of tumor invasion as well as nodal involvement.
Endoscopic ultrasound diagnostic accuracy is operator dependent and ranges from 57 to 88% for T staging in 30 to 90% for N staging.
Laparoscopy can be used to detect non resectable or disseminated disease and spare patients a full laparotomy.
CT abdomen scans can provide information about the extent of disease for staging purposes and can detect metastases, signs of locally advanced disease and adenopathy.
PET scans not as useful as in other cancers as the primary lesion uptake is seen in only 75% of cases.
PET/CT scans are better than either PET scan or CT abdomen scans alone.
Neither mucus containing tumors, diffuse type tumors or poorly differentiated scirrhous tumors visualize well with PET scans.
CEA and CA-19-9 elevated in 40-50% of patients with disseminated disease.
Adjuvant chemotherapy with 5FU, adriamycin and mitomycin after surgical resection of no statistical benefit compared to surgery alone.
For patients with localized gastric cancer, surgery is the mainstay of curative treatment in western countries.
The addition of perioperative chemotherapy has become the standard of care on the basis of studies showing that the addition of perioperative epirubicin, cisplatinum, and fluoruracil/ leukovorin, oxaliplatin, and docetaxel improved overall survival.
The addition of preoperative chemoradiotherapy to preoperative chemotherapy did not improve overall survival compared with perioperative chemotherapy alone among patients with resectable gastric and gastroesophageal junction adenocarcinoma (Leon’s T).
Postoperative radiation and 5FU/Leucovorin increases median survival by 15 months compared to surgery alone.
Preoperative and postoperative adjuvant therapy for high risk patients associated with approximately 10% increase in survival rate-includes various agents-5FU, Cisplatin, Epirubicin, capecitabine and oxaliplatin.
Pre and postoperative Docetaxel, cisplatin and 5FU has significant better time to progression, and overall survival than cisplatin plus 5FU alone.
Systemic therapy can provide palliation of symptoms, improve survival, and enhance quality life and patience with local advance to her meds the gastric cancer.
Systemic therapy can provide palliation of symptoms, improve survival, and enhance quality life and patients with locally advanced or metastatic gastric cancer.
First line therapy includes:
Docetaxel and paclitaxel response rates 11-24%.
Docetaxel, cisplatinum and 5FU is demonstrated a high response rate, prolonged survival in unresectable or metastatic gastric or gastroesophageal junction adenoarcinoma.
Combinations of a fluoro pyrimidine and a platinum agent are preferred first line treatments, along with FOLFOX/FOLFIRI.
Triplet regimens with taxane or epirubicin basedtherapy have shown efficacy and patient with locally advanced for metastatic gastric cancer.
A combination of FOLFOX with nab-taclitaxel with showed overall objective responsive 43% in patients with gastric advanced adenocarcinoma.
An estimated 5 to 70% of gastric cancer are positive for HER2.
Approximately 22% of patients with inoperable locally advanced, recurrent, or metastatic adenocarcinoma of the stomach of gastroesophageal junction have tumors specific overexpression of the HER2 gene or amplified HER2.
HER2 varies with histological subtype: the highest occurring in the intestinal subtypes and the lowest in diffuse mixed subtypes.
HER2 overexpression is more common in gastroesophageal junction cancer than those located in the gastric body.
Adding trastuzumab to standard chemotherapy for advanced disease increased survival by 26% in patients that expressed high levels of HER 2 protein compared to chemotherapy alone.
GATSBY study of trastuzumab emtansine in advanced HER2 positive gastro esophageal cancer progressing on a platinum and a fluoropyrimidine had a 20% objective response rate.
Studies show that the use of trastuzumab Deruxetan as a second line therapy in patients with HER2 positive advanced gastric or gastroesophageal junction cancer.
Ramucirumab an agent indicated for the treatment of patients with advanced or metastatic, gastric or gastroesophageal junction adenocarcinoma with disease progression on or after fluoropyrimidine or platinum containing chemotherapy.
Ramucirumab is an anti-angiogenic therapy under the trade name Cyramza.
Ramucirumab overall survival as a single agent in patients who have failed previous chemotherapy is 5.2 months compared to placebo of 3.8 months, a 37% % increase in median overall survival.
PD1 inhibitors have efficacy.
Pembrolizumab approved for patients with recurrent, locally advanced or metastatic gastric or gastrointestinal junction adenocarcinoma, whose tumors express programmed cell death ligand PD-L1.
In gastric and gastroesophageal junction (GEJ) cancers, positive for PD-L1, treatment with the PD-1 inhibitor pembrolizumab chemotherapy offered comparable survival with fewer side effects, according to results of a phase 3 randomized clinical trial (Tabernero).
These findings make pembrolizumab a preferred therapy for many patients, particularly in light of its improved safety profile versus chemotherapy.
Among patients with unresectable, locally, advanced or metastatic gastric/gastroesophageal junction, adenocarcinoma treated with first line chemotherapy and sintilimab (a PD-L1inhibitor) significantly improved overall survival for all patients and for patients with a combined positive PD-L1 score of 5 or more compared with placebo.
Treatment with perioperative capecitabine, oxiplatinum, and pembrolizumab, have encouraging activity and acceptable toxicity in patients with gastric and gastroesophageal junction adenocarcinoma.
Perioperative durvalumab plus (FLOT) 5FU, leucineovorin, oxiplatin and docetaxil led to significantly better event free survival outcomes than FLOT alone among participants with resectable gastric or gastroesophageal junction adenocarcinoma.
Trifluridine/tipracil approved for the treatment of adults with metastatic gastric or gastroesophageal junction (GEJ)
