 Cranioplasty is a surgical operation on the repairing of cranial defects caused by previous injuries or operations, such as decompressive craniectomy.
Cranioplasty is a surgical operation on the repairing of cranial defects caused by previous injuries or operations, such as decompressive craniectomy.
It is performed by filling the defective area with a range of materials, usually a bone piece from the patient or a synthetic material.
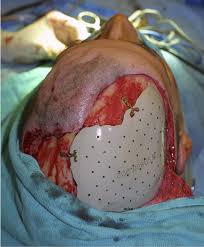
Cranioplasty is carried out by incision and reflection of the scalp.
The temporalis muscle is reflected, and all surrounding soft tissues are removed, thus completely exposing the cranial defect.
The cranioplasty flap is placed and secured on the cranial defect. The wound is then sealed.
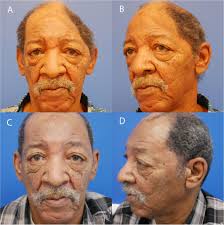
The procedure is performed for both cosmetic and functional purposes.
Cranioplasty can restore the normal shape of the skull and prevent other complications caused by a sunken scalp.
Cranioplasty is a risky operation, with potential risks such as bacterial infection and bone flap resorption.
The operation provides structure to the skull and protection to the brain from physical damage.
The surgery restores regular cerebrospinal fluid (CSF) and cerebral blood flow dynamics, along with normal intracranial pressure.
Cranioplasty may improve neurological function in some individuals.
It reduce the occurrence of headaches caused by injury or previous surgery.
The time between a craniectomy and a cranioplasty is usually between 6 months to a year, while others stated that the two operations should be more than a year apart.
Sufficient time is required for the recovery of the incision from the previous operation, as well as to clear any systemic or cranial infections.
A greater infection rate is associated with early cranioplasty due to interruption of wound healing, as well as an increased incidence of hydrocephalus.
On the contrary, evidence exists of early cranioplasty limiting complications caused by changes in cerebral blood flow and abnormal cerebrospinal fluid hydrodynamics.
Other researchers reported no significant difference in infection rate with different operational timings.
Contraindications for cranioplasty include the presence of bacterial infection, brain swelling, and hydrocephalus.
The patient is anaesthetised and an incision is made following the incision of the previous operation.
The scalp is reflected to completely reveal the cranial defect.
The cranioplasty material is placed on the defect and is fixed to the surrounding skull with standard titanium plate and screws.
CSF may be drained from the brain to reduce herniation risk.
Small holes may be drilled on the bone graft or the prosthesis to prevent the accumulation of fluid under the repaired defect.
Soft tissues, temporalis, and the scalp are then fixed back in place.
Special considerations to children undergoing cranioplasty to accommodate for their growing cranium.
Autologous bone grafts are the most preferred materials for pediatric cranioplasty, however, they may be unavailable or unsuitable in certain occasions.
Synthetic materials are used for pediatric cranioplasty when the use of autografts is not available or not recommended.
Hydroxyapatite for childhood cranioplasty allows the expansion of cranium for children and its ability to be moulded smoothly, but is less commonly used than autografts due to its brittle nature, high infection rate, and poor ability to integrate with the human cranium.
Bilateral cranioplasties are more prone to complications compared to unilateral cranioplasties in children: larger scalp wound area, a higher volume of blood loss, and the higher complexity and duration of the operation.
Cranioplasty is an operation with a complication risk ranging from 15 to 41%.
Male patients and older patients are groups with higher rates of complication.
Complications occurring after cranioplasty include:
bacterial infection, bone flap resorption, wound dehiscence, hematoma, seizures, hygroma, and cerebrospinal fluid (CSF) leakage.
The risk of bacterial infections with cranioplasty ranges from 5 to 12.8%.
The materials used for the operation affects the infection rate.
Using titanium, whether being custom-made or using a mesh, is associated with a lower infection rate.
Methyl methacrylate and autologous bone is associated with a higher infection rate.
A risk factor for bacterial infection is the location of the operation: Bifrontal cranioplasties are associated with significantly higher infection rates and higher rates for reoperation.
Other risk factors for infection: previous infections, contact between sinuses and operation site, loss of blood supply in the scalp, previous operations, and type of injury.
Bone resorption is a complication of cranioplasty with a complication rate of 0.7-17.4%,and occurs when the autologous graft does not have blood supply due to devitalisation, or when scar tissues or soft tissues remain on the edge of the cranial defect during cranioplasty.
Pediatric patients have a higher risk of resorption, up to 50%, and is more likely to occur when their cranioplasty is carried out over 6 weeks from their previous operation.
Fragmented bone and large bone flaps (>70 cm2), are associated with a higher resorption rate.
Titanium can be used as a plate, a mesh, and be 3D printed as a porous form.
Titanium is non-ferromagnetic and non-corrosive, making the host free from inflammatory reactions.
It is strong, preventing patients from trauma.
The use of titanium is associated with a lower infection rate.
Disadvantages of titanium include its high cost, poor malleability, and disruption to CT scan images.
Hydroxyapatite is a compound of calcium phosphate being arranged in a hexagonal structure.
Hydroxyapatite bonds well chemically with bones and has little inflammatory reaction by the host, has good osteointegration,
can be expanded, can be moulded smoothly and has appealing cosmetic results.
It is used in pediatric cranioplasty, but is brittle and has low tensile strength, and is only suitable to be used for small cranial defects.
Hydroxyapatite use is associated with a high infection rate.
Hydroxyapatite is often used with a titanium mesh to prevent fractures and for better osteointegration.
