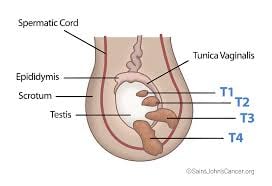
 Most common neoplasms between ages 20 and 40 years.
Most common neoplasms between ages 20 and 40 years.
Mean age at diagnosis is 33 years.
Approximately 6% of cases occur in children and adolescents younger than 18 years and 8% occur in men older than 55 years.
Estimated 10,000 cases diagnosed yearly in US.
About 500 deaths occurred from germ cell testicle tumors in 2024.
Approximately 75,000 testicular cancers are diagnosed annually with 9000 deaths per year, worldwide.
Testicle cancer incidence is currently highest among young men of European ancestry: recent registry findings reveal Hispanic individuals have an increased rate of testicular cancer among any ethnic group in the US.
In 2012 233,602 men were living with testicle cancer in the US.
Germ cell tumors compromise 95% of malignant testicle tumors, and are categorized as seminomas or nonseminomas.
The majority of testicular GCTs are type II arising from germ cell neoplasia in situ and are associated with extra copies of the short arm of chromosome 12.
GCTs develop from arrested, primordial germ cells known as genocytes that failed to differentiate into spermatogonia due to a combination of genetic and prenatal environmental factors.
Arrested gonocyte transform into total potential malignant germ cells, which are precursor for both seminoma and non-seminomatous GCT‘s.
Germ cells differentiate into seminoma, or embryonal carcinoma, which then transforms into choriocarcinoma, yolks or teratoma.
Teratomas are composed of at least two embryonic cell layers that may also undergo transformation into a secondary somatic malignancy.
In adolescence, approximately 90% of testicular germ cell tumors are non-seminomas.
Germ cell tumors are thought to arise from primordial germ cells and can develop into multiple different histologies, including seminoma, embryonal carcinoma, yolk sac tumor, teratoma, and choriocarcinoma.
When a tumor is 100% seminoma, it is referred to as a seminoma.
If any other elements are present, even if the tumor is 99% seminoma, the tumor is referred to as a nonseminoma or nonseminomatous germ cell tumor (NSGCT).
Seminomas represent 50% to 60% of testicular GCTs.
GCTs are malignancies of the primordial germ cells, which are the cells that become spermatozoa.
Most NSGCTs represent a mix of different histologies and are sometimes referred to as mixed GCTs.
GCTs are associated with the serum tumor markers alpha fetoprotein (AFP), beta human chorionic gonado- tropin (β-HCG), and lactate dehydrogenase (LDH), but none of these are pathognomonic for testis cancer or GCTs.
Seminomas do not produce AFP, and an elevated AFP thus indicates the presence of nonseminomatous elements.
Genetic and environmental factors, both in utero and during childhood are proposed to be responsible for increased incidence, which has been observed only in Caucasian males.
Family and twin studies have shown that testicular germ cell tumors have a strong inherited genetic component with an estimated heritability of 48.9%, making such tumors far more heritable than breast, ovarian, and colorectal cancers. (Litchfield K).
Testicular cancer is less common in African-Americans in the incidence among African-Americans estimated to be 1/4 that in white.
Neoplastic transformation occurs in the cells and they acquire histologic features reflecting the broad differentiation abilities of germ cells.
The initial tumorogenic event leads to the development of the germ cell tumors occurs in utero and produces the precursor lesion, the intratubular germ cell neoplasia
The intratubular germ cell neoplasia is a precursor lesion to both seminomas and non seminomatous germ cell tumors.
The intratubular germ cell neoplasia is present in testicle tissue that is adjacent to germ cell tumors in approximately 90% of cases.
Individuals with intratubular germ cell neoplasia have a 50% risk the development of testicular cancer within five years.
Intratubular germ cell neoplasia is deroved from gonocytee that have failed to differentiate into sperm and remain quiescent from the initial insult in utero until hormonal changes during pubescence.
Testicular germ cell tumors are divided into two major groups: Seminomas and nonseminomas, each accounting for 50% of cases.
These two major tumor types differ in pathogenesis, histology, clinical course and therapeutic responses.
Seminomas consist of germ cells that have been transformed and resemble gonocytes but have a differentiation block.
Testicular cancer is the most common solid tumor occurring in men between 15 and 34 years of age.
Accounts for 1% of cancer in males in the U.S.
0.4% of all US men will be diagnosed with testicle cancer in their lifetime.
Incidence has doubled in last two decades.
8000 cases per year with approximately 390 deaths.
A history of testicular cancer in the contralateral testis confers an approximately 2% risk for a second primary testicular neoplasm.
Rare in African-Americans (1.6per 100,00 population), but they experience higher grade disease and a worse survival at 5-10 years.
White patients usually present with disease at earlier stage disease compared with African-Americans, Native Americans, Hawaiians, and Hispanic patients.
Highest incidence in Denmark and lowest in the Far East.
Incidence about 5.5/100,000 population.
Bimodal age incidence with major peak in tumors have been identified to be related to mutations in the KIT gene which presumably occurred prior to divisions and arrival at the gonads.
The vast majority of testicular GCTs in adolescent and adult males are type II testicular GCTs, which arise from germ cell neoplasia in situ and are associated with extra copies of the short arm of chromosome 12, often manifesting as isochromosome 12p.5 Type II testicular GCTs.
Gains in chromosome 12p typically are found in GCT‘s.
Primary testicular GCT‘s uniformly harbor intact TP 53.
Single gene mutations are uncommon in testicular cancer.
KIT, TP 53, KRAS/NRAS,and BRAF are the genes most commonly mutated in germ cell tumors implicated in their pathogenesis.
All lesions progress through a non-invasive stage, carcinoma-in-situ, termed intratubular germ cell neoplasia unclassified.
Around puberty all in-situ lesions likely progress to seminoma and/or non-seminomatous lesions.
Gene products KIT, octamer binding transcription factor OCT3/4 and transcription factor activator protein AP-2gamma are vital in the behavior of primordial germ cells and/or in early embryonic development are expressed in-situ lesions.
Almost all patients have abnormalities on chromosome 12.
Strongest risk factor is a history of prior testicular cancer.
1-2% of patients with this lesion will develop a second primary in the contralateral testicle over time.
Risk of a second testicular cancer is 500 times the risk for the general population.
NCI-Surveillance, Epidemiology and End Results Program 15 year cumulative risk of the development of a metachronous contralateral testicular cancer is 1.9%.
Testicle ultrasound can distinguish a testicular mass from an extra testicular mass and may detect lesions not palpable on physical examination.
Testicular ultrasound can detect a testicular mass and is a sensitive and important in determining whether massesis intra or extratesticular.
Testicular ultrasound is considered the initial test of choice and is almost 100% diagnostic.
Scrotal ultrasound reveals a hypoecgoic mass and is diagnostic of testicular cancer.
Urologic evaluation is recommended when testicular lumps do not transilluminate on ultrasound or when the body of the testes cannot be palpated due to the presence of a hydrocele.
95% of masses are malignant, but ultrasound cannot distinguish benign from malignant.
Testicular lesions with ultrasound reveal hypoechoic changes.
Testicular changes of nonseminomatous tumors have hypoechoic and hyperechoic changes on ultrasound.
Cumulative 10 year risk of developing metachronous testicular cancer for patients with extragonadal germ cell tumors is 10.3%.
With extragonadal tumors of the retroperitoneum an nonseminomatous cell type there is a 14.3% 10 year risk for development of metachronous testicular cancer.
Increased risk of infertility present for men with unilateral cancers successfully treated with orchiectomy.
Increased risk include: Prior history of germ cell tumor, positive family history, cryptorchidism, testicular dysgenesis, infertility, cannabis exposure and Klinefelter syndrome.
40% of patients have subnormal sperm counts at the time of orchiectomy and 25% have such counts by 1 year.
Associated with hydrocele 20% of the time.
Inguinal lymphadenopathy not seen in the absence of an unviolated scrotum.
May have low back pain if retroperitoneal lymphadenopathy is present.
The presence of bilateral gynecomastia may be present.
With massive retroperitoneal lymphadenopathy nausea, vomiting, abdominal pain and constipation may be present.
Classified as seminoma or non-seminoma tumor.
Nonseminomatous tumors include multiple cell types such as embryonal cell, choriocarcinoma, yolk sac and teratoma.
Teratomas are considered either mature or immature, dependent on the adult-type differentiation or partial somatic differentiation that is found.
Seminoma most common histology in older population and rare in those younger than 10 years.
Mediastinal adenopathy may be associated with cough, chest pain or shortness of breath.
Spread to the lungs may be associated with dyspnea, cough, or hemoptysis.
Osseous metastases is most common in patients with seminoma.
Liver metastases are not uncommon.
Symptoms include painless mass to diffuse pain, swelling and hardness.
Most common presentation is a painless scrotal mass that cannot be separated from the testis on physical examination (90%).
About 10% of patients with testicular cancer have acute testicular pain attributable to rapid tumor growth that may cause in testicular hemorrhage, infarction, or rarely testicular torsion.
Scrotal, swelling, heaviness, discomfort, or testicular shrinkage are manifestations of testicular cancer in less than 10% of cases.
The mass may be painful and must be differentiated from epididymitis or testicular torsion.
Most often patients present with testicular discomfort or swelling suggestive of epididymitis or orchitis
Persistent tenderness or swelling or palpable abnormality warrants evaluation.
10 to 20% of patients with GCT‘s initial symptoms are related to metastatic disease.
Testicular cancer may spread to the retroperitoneal lymph nodes, and result in a palpable abdominal mass, can obstruct the ureters and cause flank pain, compress or invade the bowel, causing G.I. symptoms, impede venous blood flow, causing a varicose, or invade the spinal muscles on nerves, causing back pain, palpable neck masses may occur in patients with metastases to the supracollicular lymph nodes.
Gynecomastia occurs at 2% of men with testicle cancer due to elevated hCG secreted by the tumor or released by damage testicular germinal epithelium, resulting in increased estradiol to testosterone ratios.
Rarely testicular cancer can present with lower extremity edema compression or invasion of the inferior vena cava.
Brain metastasis or paraneoplastic encephalitis are rare manifestations.
Greater than 90% of patients with newly diagnosed testicular germ cell cancers are cured.
A radical inguinal orchidectomy is required for all suspected testicle tumors.
Germ cell tumors tend to spread it in orderly and predictable fashion to defined lymphatic areas in the retroperitoneum.
Staging should be determined by CT of the chest, abdomen, and pelvis and measurement of tumor markers for a germ cell tumors including alpha-fetoprotein and human chorionic gonadotropin.
LDH levels should be checked on the day the chemotherapy is initiated as it is an indicator of the bulk of the disease, but is not independently used as a tumor marker or prognostic criterion.
Retroperitoneal lymph node dissection provides pathologic staging and therapeutic benefit by surgically removing microscopic nodal disease.
Clinical stage I and IIA patients with normal markers who have low-volume nodal metastases have a low incidence of relapse and can be managed by observation only if compliance can be assured.
Cure rates for stage I and low-volume stage II testis tumor patients approach 100%.
5 year survival rates 98%
Poor prognostic patients have 45-55% long-term survival rates.
80% percent of patients with metastatic disease are cured.
Presently GCT have a five year survival rate of 99%, 92%, and 85% for stages I,II, and III, respectively(2025).
The presence of malignant germ cell tumor elements in residual masses confers a relatively poor prognosis, even if the masses are completely resected.
If, at the end of chemotherapy, markers are within normal limits but significant respectable residual masses remain, they are usually resected.
Resection of residual teratoma or viable cancer after chemotherapy improves event-free and overall survival.
Mature teratoma of the testicle expresses high levels of retinoblastoma protein, pRB.
Most treat advanced lesions with systemic chemotherapy followed by postchemotherapy retroperitoneal lymph node dissection with findings of fibrosis and or necrosis in 40-50% of patients, teratoma in 35-40% of patients and viable tumor in 10-15% of patients-therefore up to 50% undergo surgery without benefit.
Post chemotherapy retroperitoneal lymph node dissection is often necessary to resect teratoma and viable residual germ cell tumors, which comprise 40% and 15% of selected specimens respectively, while necrosis and fibrosis account for the remainder of tissue.
In the patients with small residual retroperitoneal masses after chemotherapy with a diameter of 1 cm or less, 20-25% will harbor teratoma and approximately 5% we’ll have viable germ cell tumor- management of such patients is problematic as to provide surveillance or retroperitoneal lymph node dissection.
Serum levels of beta HCG and alpha-fetoprotein (AFP) are elevated in about 80-85% of patients with extensive germ cell tumors.
Elevated beta hCG levels were seen with seminomatous and nonseminomatous tumors.
Nonseminomatous testicle tumors or more clinically aggressive lesions, And when both types of elements are present, management follows at for non-seminoma.
A diagnosis of seminoma is rstricted to those with pure seminoma histology and a normal level of AFP.
AFP is produced by nonseminomatous cells such as embryonal carcinoma sac tumor and seen at any stage of disease.
Tumor markers are assessed before orchiectomy.
Serum tumor markers should be determined before and after treatment and throughout the follow-up period as they are useful for monitoring stage of nonseminomatous disease.
Serum tumor markers are useful in monitoring metastatic seminomas, as elevated markers are early signs of relapse.
Serum tumor markers for B-hCG and alpha-fetoprotein should decrease after surgery and or chemotherapy according to half-life times of 36 hours for B-hCG and 6 days for alpha-fetoprotein.
Elevated serum alpha-fetoprotein levels present in 10-20% of patients with clinical stage I disease, 20-40% of low-burden clinical stage II disease, and 40-60% of high-burden disease.
Beta-hCG elevations of approximately 10-20% of clinical stage I patients, 20-30% of patients with low-burden clinical stage II disease and 40% of patients with advanced disease.
Approximately 15-25% of patients with advanced pure seminoma have increased beta-hCG levels related to syncytial trohpoblastic elements within the tumor.
Alpha-fetoprotein can be normal in 20% of patients.
Compared to AFP and hCG LVH is a less specific marker of tumor activity.
AFP elevations associated with non-eminomatous disease.
In a testicular seminoma with an elevated AFP present it is generally assumed an undetected focus of nonseminomatous disease is present.
Increased levels of LDH observed in 60% of patients with advanced nonseminomatous germ cell tumors and 80% of patients with advance seminomas.
Inguinal orchiectomy is the primary treatment for most patients with a testicular mass.
Staging is based on hCG, LDH, and AFP values and determining the status of retroperitoneal lymph nodes, the presence of lung, brain or bone metastases.
The international Germ Cell Cancer Consensus Group (IGCCCG) defines prognosis on features such as extensive disease and levels serum tumor markers after orchiectomy.
An abdominal/pelvic CT scan is performed to assess retroperitoneal nodes.
A CT of the thorax as indicated if abdominal pelvic CT shows retroperitoneal adenopathy or chest x-ray shows abnormal results.
MRI of the brain or bone scan are done only in cases with suspicious metastases.
Surveillance for postorchiectomy stage I include serum tumor markers every 3 to 6 months for the first year, every 6 to 12 months for years 3 and 3 and annually thereafter.
Surveillance for postorchiectomy stage I include abdominal/pelvic CT every 3,6, and 12 months for the first yea, every 6-12 months for years 2 and 3, and then every 12 to 24 months for years 4 and 5.
Initial relapses in the lung have not been reported in patients with stage I seminoma managed by active surveillance and routine chest imaging is only indicated for patients with thoracic symptoms.
The risk of recurrence after adjuvant treatment with carboplatin or radiation resistant 0.3% annually in follow-up in such patients includes serum tumor markers every 6-12 months for the first 2 years and annually thereafter.
Meta-analysis of 2466 patients treated with either radiation or carboplatinum for stage I disease, recurrence rarely occurred after more than 3 years, involving only 0.2 patients.
Stage I seminoma patients treated with radiotherapy or carboplatinum are recommended to have abdominal pelvic CT scans annually for 3 years and chest radiographs obtained only one clinically indicated.
Pure seminoma stage IS refers to persistent elevation of serum tumor markers after orchiectomy and is rare and usually is evidence of metastatic disease.
Platinum, Velban and bleomycin (PVB) achieves 50-60% long term disease free survival in patients with high risk or metastatic disease.
BEP (bleomycin, etoposide and platinum) is less toxic and associated with higher cure rates than PVB in disseminated testicle germ cell tumors.
20-30% of patients receiving first line chemotherapy for disseminated disease require some form of salvalge therapy.
Salvage therapy for testicular cancer includes a platinum based treatment with drugs not previously used including ifosfamide, vinblastine or paclitaxel.
For patients with refractory disease standard does chemotherapy is rarely curative.
Salvage therapy for testicular cancer also includes high-dose chemotherapy with autologous hematopoietic stem cell rescue.
Survivors have an increased risk of cardiovascular disease following treatment with chemotherapy and radiation, or both, compared to patients treated with orchidectomy alone.
Long-term testicular cancer survivors of cisplatinum based chemotherapy have higher risk of hypercholesterolemia, obesity, hypothalamic-pituitary-gonadal axis dysfunction, hyperinsulinemia, metabolic syndrome and cardiovascular disease.
1.7 fold increase in risk of cardiovascular disease in 2707 testicular cancer patients associated with cisplatinum based chemotherapy (Belt-Dusebout).
Carboplatin-based combination chemotherapy is less effective than cisplatin-based combinations in randomized trials of advanced germ cell tumors.
