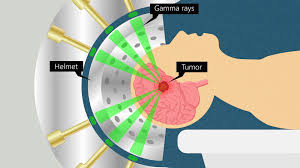
 Defined as external beam radiation therapy to deliver precise high-dose radiation to an extra cranial target within the body.
Defined as external beam radiation therapy to deliver precise high-dose radiation to an extra cranial target within the body.
Stereotactic ablative radiotherapy delivers very large hypofractionated doses of highly conformal radiotherapy to tumor targets with the aid of onboard imaging.
It utilizes either a single or small number of fractions.
Results in high target dose and steep dose gradients beyond the target.
Ultra-high doses per fraction of radiation are applied in the range of 10-20 Gy per fraction.
Designed to irradiate tumors with biologically effective doses of at least 100 Gy.
Treats lesions by converging a large number of high dose diagnosing radiation beams to target site.
Has a high conformal dose distribution, with a sharp dose-gradient fall off outside of the target.
Common treatment plans comprised 3-5 fractions of 10-20 Gy during 1-2 weeks.
Stereotactic radiosurgery is generally limited to lesions smaller than 4 cm in diameter.
Effects on adjacent normal tissues is minimalized because of multiple beam entrance and exit directions.
Stereotactic body radiation therapy well tolerated for primary or metastatic tumors of the chest that are not within 2 cm of the major airways.
Local control for thoracic lesions 85-95% at 2-3 years.
High doses in a few factions requires small field margins around a target thoracic lesion and utilizes sophisticated image guidance and adjustment for motion.
Central zone surrounding the proximal bronchial tree contains structures that appear more sensitive to the remaining lung parenchyma
Can be utilized for a variety of early-stage cancers and for the treatment of patients with metastatic disease.
Placement of fiducials are required with the CyberKnife Stereotactic Radiosurgery System.
CyberKnife Stereotactic Radiosurgery System accounts for respiration motion by tracking fiducials, usually four or more, during treatment and adjusts the trajectory of the radiation beam following the fiducials.
Some systems rely on respiratory gating, delivering radiation only during certain phases of respiration or utilize abdominal compression, limiting diaphragmatic motion with respiration and do not require fiducials.
In a study of 48 consecutive patients placement of pulmonary fiducials was associated with 33% incidence of pneumothorax and 12.5% of the patients needed the placement of a chest tube.
Pacement of fiducials at the same time of percutaneous lung biopsy associated with a higher risk of pneumonitis.
SRT protocols administered biologically effective doses of equal or greater than 100 Gy, which confers superior tumor kill compared to the 70-80 Gy
administered with conventional radiation.
SRT In patients with stage I non-small cell lung cancer 3 and five year local control rates with approximately 100 Gy are 95% and 84%, respectively.
A randomized control trial compared lung SBRT with hypo fractionated conventional radiotherapy, found no differences in local control for stage one non-small cell lung cancer (LUSTRE trial).
SRT ablative therapy is an established treatment for NSCLC, that is medically inoperable, but early stage, and is in establish therapy for pulmonary oligometastasis from various primaries.
SRT to the lung is associated with rib fractures and pneumonitis affecting up to 5% of patients.
Stereotactic body radiation for unresectable pancreatic cancer in a Stanford University study of 25 Gy 1 fraction plus adjuvant gemcitabine associated with the new survival of 12.7 months (Schellenberg D et al).
Stereotactic body radiation for unresectable pancreatic cancer with 30 Gy with 3 fractions plus gemcitabine had a median survival of 6 months at the San Bartolo Hospital (Polistina F et al).
Stereotactic body radiation for unresectable pancreatic cancer by the Beth Israel-Deaconess Hospital treated with 24-36 Gy over three fractions with gemcitabine was associated with a median survival of 14.3 months (Mahadevan A et al).
Stereotatic body radiotherapy allows delivery of focal ablative dose to hepatic metastases while sparing the normal hepatic tisue and surrounding organs at risk.
With stereotactic radiotherapy between one and five large individual daily fractions of radiotherapy are administered target only to hepatic metastases and spare normal liver.
Stereotactic radiotherapy is effective in controlling lesions that are larger in diameter than those with radioablation and is affected in controlling lesions adjacent to large blood vessels where radio frequency ablation is ineffective in these lesions.
Risk of radiation induced necrosis in normal brain tissue surrounding target lesion increases with the size of the target volume, with escalation when the volume of normal brain is exposed to a single dose of 12 Gy in the range of 5-10 cm3 or more (Lawrence YR et al).
Stereotactic radiosurgery of the surgical cavity in patients who have had complete resection of one, two, or three brain metastases significantly lowers local recurrence compared to observation alone, and the use of SRS after brain metastasis resection could be an alternative to whole-brain radiotherapy (Mahayana A).
Patients with with 5 to 20 brain metastases receiving stereotactic radiosurgery experience a lower symptom severity and less interference with daily functioning than patients who receive hippocampal avoidance whole brain radiation.
Stereotactic radio surgery has replaced whole brain radiation therapy as the standard of care for patients with limited brain metastasis from most tumors and safety and efficacy is suggested for as many as 10 targetable lesions.
Stereotactic body radiation (SBRT) to the chest wall tissues peripheral to the lung that receives more than 30 Gy in 3-5 fractions in a volume exceeding 30 cm3, the risk for chest wall pain or rib fracture escalates substantially.
SBRT using image guidance can deliver high dose radiation precisely, with a steep dose gradient at the interface between spinal cord and tumor in spinal cord metastases, creating a therapeutic window by lowering the risk for spinal cord myelopathy.
In spinal metastases SBRT can be used instead of surgery, allowing patients to avoid perioperative risk factors such as bleeding infection and anesthesia.
In young patients with residual and/or progressive benign or low-grade brain tumors requiring radiotherapy for long term tumor control, stereotactic radiation therapy compared with conventional radiation achieved superior neurocognitive and neuroendocrine functional outcomes over five years without compromising survival (Jalali R).
The use of stereotactic ablative radiotherapy in patients with oligometastasis is associated with less than 5% grade three or higher toxic effects.
It is a non-invasive definitive treatment option for primary renal cell carcinoma, where ablative doses of radiation or delivered across one to five sessions using advanced technology.: a systemic review and meta-analysis show an estimated local control of 97.2% for renal cancer.
Five fraction SBRT is non-inferior to control radiotherapy with respect to biochemical or clinical failure and may be an efficacious treatment option for patients with low to intermediate risk localized prostate cancer.
Postradical prostatectomy chemical recurrence prostate cancer treated with SBRT is well tolerated with no measurable difference declines in urinary or bowel patient reported outcomes compared with conventional fractionated radiotherapy.
