 The sarcomere is the smallest functional unit of striated muscle tissue.
The sarcomere is the smallest functional unit of striated muscle tissue.
Skeletal muscles are composed of tubular muscle cells called muscle fibers or myofibers.
Muscle fibers contain numerous tubular myofibrils.
Myofibrils are composed of repeating sections of sarcomeres, which appear under the microscope as alternating dark and light bands.
Sarcomeres are composed of long, fibrous proteins as filaments that slide past each other when a muscle contracts or relaxes.
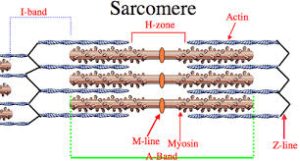
A sarcomere is the repeating unit between two Z-lines.
The costamere is a different component that connects the sarcomere to the sarcolemma.
The sarcomere has two important proteins: myosin, which forms the thick filament, and actin, which forms the thin filament.
Myosin has a long, fibrous tail and a globular head, which binds to actin.
The myosin head also binds to ATP, which is the source of energy for muscle movement.
Myosin can only bind to actin when the binding sites on actin are exposed by calcium ions.
Actin molecules are bound to the Z-line, which forms the borders of the sarcomere.
Other bands appear when the sarcomere is relaxed.
The myofibrils of smooth muscle cells are not arranged into sarcomeres.
Muscle contraction is based on sliding filament theory.
The sarcomeres give skeletal and cardiac muscle their striated appearance.
In electron micrographs of cross-striated muscle, the Z-line appears in between the I-bands as a dark line that anchors the actin myofilaments.
Surrounding the Z-line is the region of the I-band isotropic).
I-band is the zone of thin filaments that is not superimposed by thick filaments of myosin.
Following the I-band is the A-band (anisotropic).
An A-band contains the entire length of a single thick filament, and contains both thick and thin filaments.
Within the A-band is a paler region called the H-zone, which has a lighter appearance under a polarization microscope.
H-band is the zone of the thick filaments that has no actin.
Within the H-zone is a thin M-line in the middle of the sarcomere formed of cross-connecting elements of the cytoskeleton.
Actin filaments, the thin filaments, are the major component of the I-band and extend into the A-band.
Myosin filaments, the thick filaments, are bipolar and extend throughout the A-band, and are cross-linked at the centre by the M-band.
The giant protein titin, also known as connectin, extends from the Z-line of the sarcomere, where it binds to the thick filament myosin, to the M-band, where it is thought to interact with the thick filaments.
Titin is the biggest single highly elasticated protein found in nature.
It provides binding sites for numerous proteins and is a blueprint for the assembly of the sarcomere.
Another giant protein, nebulin, is hypothesised to extend along the thin filaments and the entire I-Band, and
is thought to act as a molecular ruler along for thin filament assembly.
Actin filaments and titin molecules are cross-linked in the Z-disc via the Z-line protein alpha-actinin.
The M-line also binds creatine kinase, which facilitates the reaction of ADP and phosphocreatine into ATP and creatine.
Actin and myosin filament interaction in the A-band of the sarcomere is responsible for the muscle contraction.
The tropomyosin protein covers the myosin-binding sites of the actin molecules in the muscle cell.
For a muscle cell to contract, tropomyosin must be moved to uncover the binding sites on the actin.
Calcium ions bind with troponin C molecules throughout the tropomyosin protein, and alter the structure of the tropomyosin, forcing it to reveal the cross-bridge binding site on the actin.
The concentration of calcium within muscle cells is controlled by the sarcoplasmic reticulum.
The sarcoplasmic reticulum is a form of endoplasmic reticulum in the sarcoplasm.
The motor neuron releases the neurotransmitter acetylcholine, which travels across the neuromuscular junction to stimulate muscle cells.
Acetylcholine binds to a post-synaptic nicotinic acetylcholine receptor.
A change in the receptor conformation allows an influx of sodium ions and initiation of a post-synaptic action potential.
The action potential then travels along T-tubules until it reaches the sarcoplasmic reticulum.
The depolarized membrane activates voltage-gated L-type calcium channels, present in the plasma membrane.
The L-type calcium channels are in close association with ryanodine receptors on the sarcoplasmic reticulum.
The L-type calcium channel receptors release calcium ion from the sarcoplasmic reticulum.
The outflow of calcium allows the myosin heads access to the actin cross-bridge binding sites, permitting muscle contraction.
The outflow of calcium allows the myosin heads access to the actin cross-bridge binding sites, permitting muscle contraction.
When calcium ions are pumped back into the sarcoplasmic reticulum, muscle contraction ends allowing the contractile apparatus and, thus, muscle cell to relax.
Upon muscle contraction, the A-bands do not change their length: the I-bands and the H-zone shortens, causing the Z lines to come closer together.
At rest, the myosin head is bound to an ATP molecule in a low-energy configuration and is unable to bind sites on the actin.
The myosin head can hydrolyze ATP into adenosine diphosphate (ADP), and a portion of the energy released in this reaction changes the shape of the myosin head and promotes it to a high-energy configuration.
By binding to the actin, the myosin head releases ADP and an inorganic phosphate ion, changing its configuration back to one of low energy.
The myosin remains attached to actin in a state known as rigor, until a new ATP binds the myosin head.
This binding of ATP to myosin releases the actin by cross-bridge dissociation.
The ATP-associated myosin is ready for another cycle, beginning with hydrolysis of the ATP.
The A-band is visible as dark transverse lines across myofibers.
The I-band is visible as lightly staining transverse lines, and the Z-line is visible as dark lines separating sarcomeres at the light-microscope level.
Muscle cells can only store enough ATP for a small number of muscle contractions.
Muscle cells also store glycogen.
Most of the energy required for muscle contraction is derived from phosphagens: creatine phosphate, is used to provide ADP with a phosphate group for ATP synthesis.
The overlap of actin and myosin proteins gives rise to the length-tension curve: sarcomere force output decreases if the muscle is stretched so that fewer cross-bridges can form or compressed until actin filaments interfere with each other.
The length of the actin and myosin filaments that makeup a sarcomere length, affects force and velocity.
Longer sarcomeres have more cross-bridges and thus more force, but have a reduced range of shortening.
