 Neurobiological affects of physical exercise occur to the brain structure, brain function, and cognition.
Neurobiological affects of physical exercise occur to the brain structure, brain function, and cognition.
Consistent daily aerobic exercise may induce improvements in certain cognitive functions, neuroplasticity and behavioral plasticity.
Aerobic exercise induces transient effects on cognition after a single exercise session and persistent effects on cognition following consistent exercise over the course of several months.
People who regularly perform aerobic exercises have greater scores on neuropsychological function and performance tests measuring certain cognitive function, such as attention control, inhibitory control, cognitive flexibility, working memory, declarative memory, spatial memory, and information processing.
Long-term effects of aerobic exercise may include increased neuron growth, increased neurological activity and signaling of Fos and BDNF, improved stress coping, enhanced cognitive control of behavior, improved declarative, spatial, and working memory, and structural and functional improvements in brain structures and pathways associated with cognitive control and memory.
Exercise’s effects on cognition may affect academic performance in children and college students, improve adult productivity, preserve cognitive function in old age, preventing or treating certain neurological disorders, and improving overall quality of life.
Aerobic exercise has both short and long term effects on mood and emotional states by promoting positive affect, inhibiting negative affect, and decreasing the biological response to acute psychological stress.
Aerobic exercise affects both self-esteem and overall well-being including sleep patterns, with consistent, long term participation.
Regular aerobic exercise may improve symptoms associated with central nervous system disorders and may be used as adjunct therapy.
Evidence exists that exercise treatment efficacy for major depressive disorder and attention deficit hyperactivity disorder.
Guidelines for mild cognitive impairment recommend regular exercise, two times per week, to individuals who have been diagnosed with this condition.
Exercise may be useful as an adjunct therapy for the treatment and prevention of drug addictions.
The use of exercise as an adjunct therapy for certain neurodegenerative disorders, particularly Alzheimer’s disease and Parkinson’s disease is recommended.
Regular exercise may be associated with a lower risk of developing neurodegenerative disorders.
Neuroplasticity is the process by which neurons adapt to a disturbance over time, and most often occurs in response to repeated exposure to stimuli.
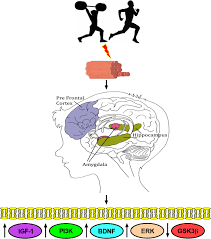
Aerobic exercise increases the production of neurotrophic factors-BDNF, IGF-1, VEGF, which mediate improvements in cognitive functions and various forms of memory by promoting blood vessel formation in the brain, adult neurogenesis, and other forms of neuroplasticity.
Consistent aerobic exercise over a period of several months induces improvements in executive functions and increased gray matter volume in nearly all regions of the brain,, with the most marked increases occurring in brain regions that give rise to executive functions.
The brain structures that show the greatest improvements in gray matter volume in response to aerobic exercise are the prefrontal cortex, caudate nucleus, and hippocampus, with less significant increases in gray matter volume in the anterior cingulate cortex, parietal cortex, cerebellum, and nucleus accumbens.
The prefrontal cortex, caudate nucleus, and anterior cingulate cortex are among the most significant brain structures in the dopamine and norepinephrine systems that give rise to cognitive control.
Exercise-induced increases in gray matter neurogenesis volume in the hippocampus is associated with measurable improvements in spatial memory.
Higher physical fitness scores, as measured by VO2 max, are associated with better executive function, faster information processing speed, and greater gray matter volume of the hippocampus, caudate nucleus, and nucleus accumbens.
Consistent aerobic exercise increases gray matter volume in nearly all regions of the brain, with more pronounced increases occurring in brain regions associated with memory processing, cognitive control, motor function, and reward.
The most prominent gains in gray matter volume with aerobic exercise are seen in the prefrontal cortex, caudate nucleus, and hippocampus, which support cognitive control and memory processing, among other cognitive functions.
The prefrontal cortex, the hippocampus, and the cingulate cortex become more functionally interconnected in response to consistent aerobic exercise.
Marked improvements in prefrontal and hippocampal gray matter volume occur in healthy adults that regularly engage in medium intensity exercise for several months.
Other regions of the brain that demonstrate moderate or less significant gains in gray matter volume during neuroimaging include the anterior cingulate cortex, parietal cortex, cerebellum, and nucleus accumbens.
Regular exercise has been shown to counter the shrinking of the hippocampus and memory impairment that naturally occurs in late adulthood.
Sedentary adults over age 55 show a 1–2% decline in hippocampal volume annually.
Regular aerobic exercise increased the volume of the left hippocampus by 2.12% and the right hippocampus by 1.97% over a one-year period.
Aerobic exercise has also been shown to induce growth in the white matter tracts in the anterior corpus callosum, which normally shrink with age.
Brain structures that show exercise-induced increases in gray matter volume include:
Caudate nucleus – responsible for stimulus-response learning and inhibitory control; implicated in Parkinson’s disease and ADHD
Cerebellum – responsible for motor coordination and motor learning
Hippocampus – responsible for storage and consolidation of declarative memory and spatial memory
Nucleus accumbens – responsible for wanting or desire, the form of motivation associated with reward, and positive reinforcement, implicated in addiction.
Parietal cortex – responsible for sensory perception, working memory, and attention
Prefrontal and anterior cingulate cortices – required for the cognitive control of behavior, particularly: working memory, attentional control, decision-making, cognitive flexibility, social cognition, and inhibitory control of behavior; implicated in attention deficit hyperactivity disorder (ADHD) and addiction
Regular exercise over a period of several months has been shown to persistently improve numerous executive functions and several forms of memory attentional control, processing speed, cognitive flexibility, task switching, inhibitory control, nondeclarative memory, and spatial memory.
In healthy young and middle-aged adults, the effect sizes of improvements in cognitive function are largest for executive functions and small to moderate for aspects of memory and information processing speed.
Older adults benefit cognitively by taking part in both aerobic and resistance type exercise of at least moderate intensity.
Individuals who have a sedentary lifestyle tend to have impaired executive functions relative to other more physically active non-exercisers: A reciprocal relationship between exercise and executive functions has also been noted: improvements in executive control processes, such as attentional control and inhibitory control, increase an individual’s tendency to exercise.
One of the most significant effects of exercise on the brain is increased synthesis and expression of BDNF, a neuropeptide and hormone, resulting in increased signaling through its receptor tropomyosin receptor kinase B (TrkB).
BDNF is capable of crossing the blood–brain barrier, higher peripheral BDNF synthesis also increases BDNF signaling in the brain.
Exercise-induced increases in BDNF signaling are associated with improved cognitive function, improved mood, and improved memory.
BDNF has a role in hippocampal neurogenesis, synaptic plasticity, and neural repair.
Engaging in moderate-high intensity aerobic exercise such as running, swimming, and cycling increases BDNF biosynthesis through myokine signaling, resulting in up to a threefold increase in blood plasma and BDNF levels; exercise intensity is positively correlated with the magnitude of increased BDNF biosynthesis and expression.
Consistent exercise modestly increases resting BDNF levels as well.
Exercise as a mechanism to reduces stress since stress is closely linked with decreased levels of BDNF in the hippocampus.
The increase in BDNF levels caused by exercise helps reverse the stress-induced decrease in BDNF which mediates stress in the short term and buffers against stress-related diseases in the long term.
IGF-1 is a peptide and neurotrophic factor that mediates some of the effects of growth hormone.
IGF-1 elicits its physiological effects by binding to a specific receptor tyrosine kinase, the IGF-1 receptor, to control tissue growth and remodeling.
In the brain, IGF-1 functions as a neurotrophic factor that, like BDNF, plays a significant role in cognition, neurogenesis, and neuronal survival.
Physical activity is associated with increased levels of IGF-1 in blood serum, which is known to contribute to neuroplasticity in the brain due to its capacity to cross the blood–brain barrier and blood–cerebrospinal fluid barrier.
IGF-1 appears to be a key mediator of exercise-induced adult neurogenesis.
IGF-1 as a factor which links the body’s fitness with brain fitness.
The amount of IGF-1 released into blood plasma during exercise is positively correlated with exercise intensity and duration.
Vascular endothelial growth factor (VEGF) is a neurotrophic and angiogenic, that is, blood vessel growth-promoting signaling protein that binds to two receptor tyrosine kinases, VEGFR1 and VEGFR2, which are expressed in neurons and glial cells in the brain.
Hypoxia, or inadequate cellular oxygen supply, strongly upregulates VEGF expression and VEGF exerts a neuroprotective effect in hypoxic neurons.
Like BDNF and IGF-1, aerobic exercise has been shown to increase VEGF biosynthesis in peripheral tissue which subsequently crosses the blood–brain barrier and promotes neurogenesis and blood vessel formation in the central nervous system.
Exercise-induced increases in VEGF signaling improves cerebral blood volume and contribute to exercise-induced neurogenesis in the hippocampus.
There are persistent effects on cognition resulting from several months of daily exercise.
Acute exercise has been shown to transiently improve a number of cognitive functions.
Reviews and meta-analyses on the effects of acute exercise on cognition in healthy young and middle-aged adults have concluded that information processing speed and a number of executive functions – including attention, working memory, problem solving, cognitive flexibility, verbal fluency, decision making, and inhibitory control, all improve for a period of up to 2 hours post-exercise.
Studies conducted on children also suggested that some of the exercise-induced improvements in executive function are apparent after single bouts of exercise, while other aspects, such as, attentional control only improve following consistent exercise on a regular basis.
Runners can experience a euphoric state often-runner’s high.
Continuous exercise can produce a transient state of euphoria – an affective state involving the experience of pleasure and feelings of profound contentment, elation, and well-being – which is known as a high.
Endogenous euphoriants are responsible for producing exercise-related euphoria, specifically phenethylamine, which is an endogenous psychostimulant, β-endorphin an endogenous opioid, and anandamide, an endogenous cannabinoid.
β-Phenylethylamine, commonly referred to as phenethylamine, is a human trace amine and potent catecholaminergic and glutamatergic neuromodulator that has similar psychostimulant and euphoriant effects and a similar chemical structure to amphetamine.
Thirty minutes of moderate to high intensity physical exercise has been shown to induce an enormous increase in urinary β-phenylacetic acid, the primary metabolite of phenethylamine.
The average 24 hour urinary β-phenylacetic acid concentration among participants following just 30 minutes of intense exercise increased by 77% relative to baseline concentrations in resting control subjects.
Phenethylamine synthesis sharply increases while an individual is exercising, during which time it is rapidly metabolized due to its short half-life of roughly 30 seconds.
Phenethylamine plays a prominent role in mediating the mood-enhancing euphoric effects of a runner’s high, as both phenethylamine and amphetamine are potent euphoriants.
β-Endorphin is an endogenous opioid neuropeptide that binds to μ-opioid receptors, in turn producing euphoria and pain relief.
Exercise significantly increases the secretion of β-endorphin and that this secretion is correlated with improved mood states.
Moderate intensity exercise produces the greatest increase in β-endorphin synthesis, while higher and lower intensity forms of exercise are associated with smaller increases in β-endorphin synthesis.
A review on β-endorphin and exercise noted that an individual’s mood improves for the remainder of the day following physical exercise and that one’s mood is positively correlated with overall daily physical activity level.
Anandamide is an endogenous cannabinoid and retrograde neurotransmitter that binds to cannabinoid receptors, primarily CB1.
Anandamide produces euphoria.
It has been shown that aerobic exercise causes an increase in plasma anandamide levels.
The magnitude of this increase in production is highest at moderate exercise intensity, of about 70–80% maximum heart rate.
Increases in plasma anandamide levels are associated with psychoactive effects because anandamide is able to cross the blood–brain barrier and act within the central nervous system.
Anandamide partly mediates the short-term mood-lifting effects of exercise.
The stress hormone cortisol, is a glucocorticoid that binds to glucocorticoid receptors.
Psychological stress induces the release of cortisol from the adrenal gland by activating the hypothalamic–pituitary–adrenal axis (HPA axis).
increase in cortisol levels are associated with adaptive cognitive improvements, such as enhanced inhibitory control.
Excessively high exposure or prolonged exposure to high levels of cortisol, however, causes impairments in cognitive control and has neurotoxic effects in the human brain.
Chronic psychological stress decreases BDNF expression, which has detrimental effects on hippocampal volume and can lead to depression.
Aerobic exercise stimulates cortisol secretion in an intensity-dependent manner.
Aerobic exercise increases physical fitness and lowers neuroendocrine reactivity, reducing the biological response to psychological stress.
Exercise reverses stress-induced decreases in BDNF expression and signaling in the brain.
Exercise acts as a buffer against stress-related diseases like depression.
Glutamate, a neurochemical in the brain, is an excitatory transmitter involved in many aspects of brain function, including learning and memory.
Physical pursuits has positive effects on the mental health of children and adolescents, enhances their academic performance, boosts cognitive function, and diminishes the likelihood of obesity and cardiovascular diseases.
Establishing consistent exercise routines with regular frequency and duration is pivotal, and may support the overall physical and mental well-being of young individuals.
A meta-analysis found a positive effect of exercise in children on perceptual skills, intelligence quotient, achievement, verbal tests, mathematic tests, and academic readiness:The correlation was strongest for the age ranges of 4–7 and 11–13 years.
Aerobic exercise may briefly aid children’s executive function and also influence more lasting improvements to executive function.
Consistent aerobic exercise, especially endurance exercise prevents the development of certain drug addictions and is an effective adjunct treatment for drug addiction, and psychostimulant addiction in particular.
Consistent aerobic exercise magnitude-dependently may reduce drug addiction risk, which appears to occur through the reversal of drug-induced, addiction-related neuroplasticity.
Aerobic exercise decreases psychostimulant self-administration, reduces the relapse of drug-seeking, and induces opposite effects on striatal dopamine receptor D2 (DRD2) signaling to those induced by pathological stimulant use.
Aerobic exercise may lead to better treatment outcomes when used as an adjunct treatment for drug addiction.
Regular physical exercise, particularly aerobic exercise, is an effective add-on treatment for ADHD in children and adults, especially, when combined with stimulant medication.
The effects of regular aerobic exercise in ADHD individuals include better behavior and motor abilities, improved executive functions including :attention, inhibitory control, and planning, faster information processing speed, better memory, better overall function, reduced ADHD symptoms, better self-esteem, reduced levels of anxiety and depression, fewer somatic complaints, better academic and classroom behavior, and improved social behavior.
Exercising while taking stimulant medication augments the effect of stimulant medication on executive function.
Short-term effects of exercise are mediated by an increased abundance of synaptic dopamine and norepinephrine in the brain.
Exercise has a marked and persistent antidepressant effect in humans,believed to be mediated through enhanced BDNF signaling in the brain.
Physical exercise is effective as an adjunct treatment with antidepressant medication.
Physical exercise has marked antidepressant effects and recommended the inclusion of physical activity as an adjunct treatment for mild–moderate depression and mental illness in general.
Physical exercise improves overall quality of life in individuals with depression relative to controls.
Physical activity decrease the risk of ischemic stroke and intracerebral haemorrhage.
Exercise increases the expression of VEGF, and angiopoietin in the brain, which promote angiogenesis and neovascularization that contribute to improved blood supply to the stroke affected areas of the brain.
Exercise may affect the activation of endothelial nitric oxide synthase and production of nitric oxide (NO).
Increasing NO production may lead to improved post-stroke cerebral blood flow, ensuring a sufficient oxygen and nutrient supply to the brain.
Physical activity associated with increased expression and activation of hypoxia-inducible factor 1 alpha (HIF-1α), heat shock proteins, and brain-derived neurotrophic factor (BDNF), which play roles in promoting cell survival, neuroprotection, and repair processes in the brain following a stroke.
Exercise inhibits glutamate and caspase activities involved in neuronal death pathways.
Exercise promotes neurogenesis in the brain, reducing effects of brain infarction and edema, leading to potential improvements in neurological and functional outcomes.
Physical activity may reduce the volume of intracerebral haemorrhages.
Being physically active after stroke enhances functional recovery.
Regular physical exercise improves cognitive symptoms in individuals with mild cognitive impairment.
There is evidence for positive effects of physical exercise on cognitive function, the rate of cognitive decline, and the ability to perform activities of daily living in individuals with Alzheimer’s disease.
Higher levels of physical activity may be associated with reduced risk of dementia and cognitive decline.
Evidence suggests that physical exercise may lower the risk of Parkinson’s disease.
Strength and endurance training in people with Parkinson’s disease have positive effects lasting for several weeks.
