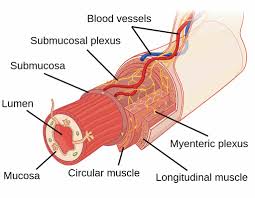
 The myenteric plexus (or Auerbach’s plexus) provides motor innervation to both layers of the muscular layer of the gut, having both parasympathetic and sympathetic input.
The myenteric plexus (or Auerbach’s plexus) provides motor innervation to both layers of the muscular layer of the gut, having both parasympathetic and sympathetic input.
It is a rare disorder (1:5000), with prevalence among males being four times that of females.
The submucous plexus provides secretomotor innervation to the mucosa nearest the lumen of the gut.
The myenteric plexus is the major nerve supply to the gastrointestinal tract and controls GI tract motility.
30% of myenteric plexus’ neurons are enteric sensory neurons, thus Auerbach’s plexus has also a sensory component.
The myenteric plexus, a part of the enteric nervous system exists between the longitudinal and circular layers of muscularis externa in the gastrointestinal tract.
The myenteric plexus is found in the muscles of the esophagus, stomach, and intestine.
Its ganglia have properties similar to the central nervous system (CNS): presence of glia, interneurons, a small extracellular space, dense synaptic neuropil, isolation from blood vessels, multiple synaptic mechanisms and multiple neurotransmitters.
The myenteric plexus originates in the medulla oblongata as a collection of neurons from the ventral part of the brain stem.
The vagus nerve then carries the axons to their destination in the gastrointestinal tract.
They contain Dogiel cells.
The myenteric plexus functions as a part of the enteric nervous system.
The enteric nervous system can and does function autonomously, but normal digestive function requires communication links between this intrinsic system and the central nervous system.
The ENS contains sensory receptors, primary afferent neurons, interneurons, and motor neurons.
The events that are controlled, at least in part, by the ENS are multiple and include motor activity, secretion, absorption, blood flow, and interaction with other organs such as the gallbladder or pancreas.
These links take the form of parasympathetic and sympathetic fibers that connect either the central and enteric nervous systems or connect the central nervous system directly with the digestive tract.
The enteric nervous system cross connections of the gut can provide sensory information to the CNS, and the CNS can affect gastrointestinal function.
Connection to the central nervous system means that signals from outside of the digestive system can be relayed to the digestive system: sight of appealing food stimulates secretion in the stomach.
The enteric nervous system utilizes of over 30 different neurotransmitters, most similar to those of the CNS such as acetylcholine, dopamine, and serotonin.
More than 90% of the body’s serotonin lies in the gut; as well as about 50% of the body’s dopamine.
Substance P is present in significant levels and may help facilitate the production of saliva, smooth muscle contractions, and other tissue responses.
Myenteric neurons can express receptors for both peptide and non-peptide (amines, amino acids, purines) neurotransmitters.
Activating this pre-synaptic the 5-hydroxytryptamine (5-HT4) receptor enhances cholinergic neurotransmission and can stimulate gastrointestinal motility.
The enteric nervous system exhibits taste receptors.
The taste receptor TAS1R3 and the taste G protein gustducin are two of the most common, and sense “sweetness” on the tongue and sense glucose in the enteric nervous system.
These receptors therefore help regulate the secretion of insulin and other hormones that are responsible for controlling blood sugar levels.
Hirschsprung’s disease is a congenital disorder of the colon in which nerve cells of the myenteric plexus in its walls are absent.
Hirschsprung’s disease is a form of functional low bowel obstruction.
Hirschsprung’s disease is due to failure of caudal migration of neuroblasts within developing bowel, with an absence of parasympathetic intrinsic ganglion cells in both Auerbach’s and Meissner’s plexuses.
The distal large bowel from the point of neuronal arrest to the anus is continuously aganglionic.
Achalasia is a motor disorder of the esophagus characterized by decrease in ganglion cell density in the myenteric plexus.
Myenteric plexi destruction has been found to be secondary to Chagas’ disease, with destruction occurs in the esophagus, intestines, and ureters.
This denervation can lead to secondary achalasia of the lower esophageal sphincter, megacolon, and megaureter.
Parkinson’s disease patients are known to experience severe constipation due to GI tract dysfunction years before the onset of motor movement complications, which characterises Parkinson’s disease.
