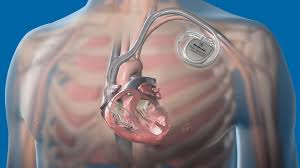
 Also known as automatic internal cardiac defibrillator (AICD).
Also known as automatic internal cardiac defibrillator (AICD).
ICD is one of the most effective therapies for the prevention of sudden death from cardiac causes.
These devices are implants, similar to pacemakers and can also perform the pacemaking function.
Transvenous ICD’s comprise, an implanted pulse generator, and one or more leads inside the heart, can terminate many ventricular tachycardias with either a high energy discharge (shock) or anti-tachycardia pacing (ATP).
ATP disrupts a ventricular tachycardia circuit by rapid pacing from a remote site, and provides treatment of potentially lethal cardiac rhythms.
Constantly monitor heart rhythm, and automatically administer shocks for various life threatening arrhythmias.
Can distinguish between ventricular fibrillation, ventricular tachycardia, and more benign arrhythmias.
With ventricular fibrillation, the device is programmed to proceed immediately to an unsynchronized shock.
Aberrant activity of ICD may lead to its firing constantly or inappropriately, and such activity is considered a medical emergency.
Inappropriate firing can deplete battery life, causes discomfort and anxiety, and trigger life threatening arrhythmias.
Defibrillation is effective if it produces a threshold voltage gradient in a large fraction of the myocardial mass, and a gradient of three to five volts per centimeter is needed in 95% of the heart.
Implantable cardioverter-defibrillators reduce cardiac mortality among patients at risk for ventricular arrhythmias.
Intraventricular cardioverter-defibrillator superior to most antiarrhythmic drugs in reducing mortality in patients with life-threatening ventricular tachyarrhythmias.
ICD reduce the risk of death, both and survivors of sudden cardiac arrest and in patients who have not had previous ventricular arrhythmias, but are known to be at increased risk.
Rates of defibrillator therapy are substantial ranging from 3 to 10% per year, in patients who have an ICD and are associated with considerable psychological distress, increased healthcare utilization, and in some cases mortality.
ICD’s decrease all cause mortality in patients at high risk for ventricular arrhythmias, especially those with reduced left ventricular systolic function, and this benefit in mortality is directly related to termination of potentially life-threatening arrhythmias.
Electrical conductive gel is used at the electrodes in order to ensure a good connection and to minimize electrical resistance, also called chest impedance which would burn the patient.
Transvenous ICD implantation may be complicated by vascular injury, cardiac perforation, pneumothorax, hemothorax, and venous obstruction.
Transvenous leads are subject to mechanical failure and serious infection resulting in lead extraction.
Gels may be either wet or solid , but the latter type has a higher risk of burn during defibrillation.
The traditional metal paddle with an insulated handle must be held in place with approximately 25 lbs of force while a shock is delivered.
Paddles allow for monitoring electrocardiographically.
Paddles are reuseable. that can be “shocked” when the patient is not in cardiac arrest, such as supraventricular tachycardia and ventricular tachycardia that produces apulse; this more-complicated procedure is known as cardioversion, not defibrillation.
Most effective treatment for patients at high risk for life threatening ventricular arrhythmias.
Common indications for an ICD include: sustained ventricular arrhythmias, or sudden cardiac arrest not due to reversible cause, chronic systolic heart failure due to ischemic or non-ischemic cardiomyopathy, an LVEF of 35% or lower, and NYHA class II or III despite optimal medical therapy for heart failure, chronic systolic heart failure due to ischemic cardiomyopathy, an LVEF of 30% or lower, and NYHA class I, despite optimal medical therapy for heart failure, and inherited cardiomyopathyies.
More than 3 million people in North America eligible for an ICD.
About 50% of patients receiving an ICD after the age of 65 years may be dead or in hospice five years out from implantation (Kramer DB).
Originally reserved for highest risk patients use expanded now for primary prevention.
Various criteria used for implantation criteria include width of QRS and T wave alterans.
Uses include congestive heart failure in younger patients with arrhythmogenic right ventricular dysplasia, long QT syndrome, and hypertrophy cardiomyopathy.
Superior to drugs in reducing mortality rates for survivors of cardiac arrest or patients at high risk for sudden cardiac death.
Indicated for primary prevention of sudden cardiac death in patients with reduced LV function and for secondary prevention of sudden death in patients with life threatening cardiac arrhythmia, including aborted sudden cardiac death.
Proven efficacy at preventing sudden cardiac death in patients with advanced systolic heart failure.
Trials of the efficacy generally have excluded patients with acute myocardial infarction, with recent coronary artery revascularization or with New York Heart Association class IV symptoms, or with newly diagnosed heart failure.
Some studies are associated with increased risk of death and worsening of heart failure with the application of ICD therapy.
ICD interventions, especially shocks, are associated with an increase in mortality and a worse prognosis in patients with heart failure.
Patients experience with intracracardiac shock is similar to those with external defibrillation.
It is unclear whether the negative effects of shocks or the natural history of extremely compromised patients accounts for adverse outcomes.
In some trials survival benefit from ICD therapy was not demonstrated in patients recovering from an acute MI patients who received ICD but at the time of coronary artery bypass graft surgery.
Among people with cardiovascular disease who had an ICD and were at higher risk for ventricular arrhythmias, treatment induced increase in plasma potassium levels led to a significantly lower risk of appropriate ICD therapy, unplanned hospitalization for arrhythmia or heart failure, or death from any cause than standard care (POTCAST Study Group).
Guidelines mandate at least a 40 day period following a myocardial infarction and ICD implantation.
ICD therapy not indicated for patients with NYHA class IV symptoms, who are not candidates for cardiac resynchronization therapy device.
Provides therapy within 5-15 seconds of arrhythmia detection and allows for defibrillation success rates approaching 100%.
Recommended as the treatment of choice in survivors of sudden cardiac death and in patients with symptomatic, sustained ventricular arrhythmias.
Recommended that survivors of cardiac arrest occurring after the first 24 hours of an acute myocardial infarction should receive an ICD for secondary prophylaxis of sudden cardiac death.
Does not confer added benefit compared with amiodarone in patients with ejection fraction >35%.
In patients with NYHA class II or III CHF and LVEF of 35% or less shock-only ICD reduces overall mortality by 23%, whereas amiodarone in this situation has no favorable effect.
In Medicare study among patients hospitalized for heart failure with an LVEF between 30 and 35%, survival at three years was better in patients who receive prophylactic ICD than in comparable patients with no ICD (Al-Khatib SM et al).
In a study of primary prevention population use in patients with moderately symptomatic heart failure a single lead ICD treatment is not associated with adverse quality of life effects over 30 months of follow-up (Mark).
Significantly reduces overall mortality in patients with left ventricular dysfunction secondary to ischemic heart disease.
Does not reduce overall mortality in patients with recent myocardial infarction ,although it does reduce death rate from arrhythmias, its effect is offset by increased deaths from nonarrhythmic cardiovascular processes.
Several large studies reveal use of ICD’s associated with a 1 year mortality rate of 2-5% compared to 10% mortality in electrophysiology directed drug treatment and 10-40% mortality in empirical antiarrhythmic drug management.
The Sudden Cardiac Death In Heart Failure Trial (SCD-HeFT) and the Multicenter Automatic Defibrillator Implantation Trial II (MADIT II) revealed that therapy with implantable cardioverter-defibrillators improves survival among patients who are at risk for sudden cardiac death but who have not previously had a sustained ventricular arrhythmia.
The Sudden Cardiac Death In Heart Failure Trial (SCD-HeFT) compared ICD treatment or amiodarone with placebo In 2521 patients with symptomatic heart failure due to ischemic or no ischemic left ventricular dysfunction, with the ICD group, demonstrating an absolute reduction in mortality of 7.2 percentage points over five years.
In a randomized, prospective, open label trial of 62,944 unselected patients with myocardial infarction- 898 with reduced left ventricular ejection fraction of 40% or less, a heart rate of 90 or more beats per minute, or both with up to 445 randomly assigned to treatment with a ICD and 453 to medical therapy alone: prophylactic ICD therapy did not reduce overall mortality among patients with acute myocardial infarction (Steinbeck G).
Approximately 125,000 implants annually in the U.S.
Approximately 30% of recipients are women.
As a consequence of use of this therapy the natural history of disorders have been modified.
In patients that have received an ICD for primary prevention and who have heart failure the occurrence of appropriate shock associated with a markedly increased risk of death (Poole J).
Defibrillator (ICD)-patients with primary ICD placement with congestive heart failure have a higher risk of death from progressive heart failure.
Implanted intravenously and can provide pacing in up to 3 cardiac chambers.
Infection of ICD uncommon and involves the generator pocket site and the epicardial leads in 90% and 10% of cases of infection, respectively.
Improves survival, especially in adults over the age of 65 years.
Multiple Automatic Defibrillator Implantation Trial (MADIT)II indicated that the presence of renal failure limits the efficacy of ICDs.
The presence of advanced heart failure symptoms have short and long-term negative prognostic factors (Bardy GH, Swindle J).
Persons with terminal illness with ICDs in place may be at increased risk for shocks due to hypoxia, electrolyte changes and heart failure.
Data from five studies involving 934 women revealed no statistical significant decrease in mortality in women with heart failure who received ICDs (Ghanbari H).
Women have a 70% higher risk of major adverse events after ICD implantation then do men (Peterson PN).
Contraindications are life expectancy 1 year or less, incessant arrhythmias, underlying significant psychiatric disease, syncope without inducible ventricular arrhythmia or structural heart disease,reversible causes of ventricular arrhythmia, and primary prevention for patients ineligible for cardiac transplantation or cardiac resynchronization therapy (Epstein AE et al).
Relative contraindication to ICD therapy include need for radiation therapy to the chest, high risk for infection, and high risk for DVT.
Dual chamber ICD’s with a lead in the right atrium and the right ventricle are indicated in patients receiving an ICD who also require pacing for brady arrhythmias, such as heart block or sinus node dysfunction.
Dual chamber devices may have benefits for patients who do not have an indication for pacing.
Dual chamber ICD’s having an enhanced rhythm detection, Which may decrease the incidence of inappropriate shocks.
There is no data to support the benefit of dual chamber implantable cardioverter defibrillator compared to a single chamber ICD for a non pacing indication, yet most implants are of dual chamber type.
In a National Cardiovascular Data Registry (NCDR) analysis found that procedural complications, that included in-hospital mortality, was at least 40% greater for dual chamber implantation compared with single chamber devices.
Prior to replacement of ICDs clinical reevaluation must be done as patients help me have changed influencing decisions about such replacement.
Guidelines for placement of an ICD device who that patient survival should be at least one year with a reasonable quality of life, and the same assessment is necessary regarding replacement of ICD.
If patients have improvement and intricately function replacement of the primary prevention ICD may not be necessary.
At the time of ICD replacement patients who have had significant complications of the device, including inappropriate shocks replacement may be cautiously considered.
Among patients receiving ICD using a long versus standard-detection interval resulted in lower rates of antitachycardia pacing and shocks, and inappropriate shocks (Gasparini, M et al, ADVANCE III trial).
Anti-tachycardia pacing is effective in reducing the number of shocks for fast ventricular tachyarrhythmias.
Studies have demonstrated that in primary prevention patients with ICD, a prolonged detection duration associated with anti-tachycardia pacing safely reduces the number of patients receiving shocks.
The use of a single burst of anti-tachycardia pacing (ATP) prior to shock in primary prevention ICD recipients, prolongs the time to first all cause ICD shock (APPRAISE ATP investigators).
In the secondary prevention trial Antiarryhtmics versus Implantable Defibrillators (AVID) the proportion of patients with an arrhythmic events, defined a sudden cardiac death, sustained ventricular arrhythmia, or ICD therapy, was 35% at three months, 53% at one year, and 68% in two years (Klein RC et al).
In the above study approximately 45% of patients received a shock within in the first year.
Meta-analysis suggests that appropriate ICD therapy occurs in up to 64% of patients and inappropriate shocks occur in up to 24% during 20-45 months of follow-up (Germano JJ et al).
As many as 1 in 3 patients with ICD receive inappropriate shocks.
Inappropriate shocks refer to the delivery of a high-voltage discharge for a reason other than a ventricular arrhythmia.
Defibrillator shocks are associated with the acute discomfort of the shock, psychological trauma, depression and posttraumatic stress disorder.
There is a paradoxical association between ICD shocks and increased mortality.
In the Multicenter Automatic Defibrillator Implantation Trial II (MADIT II) and in the Sudden Cardiac Death in Heart Failure Trial, ICD shocks were associated with a 2-5 fold increased risk of death.
While internal cardiac defibrillators reduce the incidence of sudden death in appropriate patients, most patients with such defibrillators will receive no benefit whatsoever (Uhlig K).
Physicians to receive payments from device manufacturers utilized ICD devices from those manufacturers, preferentially.
The extravascular implantable cardioverter-defibrillator (ICD) has a single lead implanted substernally to enable pause- prevention pacing, anti-tachycardia pacing, and defibrillation energy similar to that of transvenous ICDs.
The subcutaneous ICD avoids vascular risks of transvenous ICDs.
Subcutaneous ICD’s, prevent transvenous lead related issues such as infection, fracture, dislodgment, and tricuspid regurgitation.
Subcutaneous ICD is preferred in patients who have a high risk of infection, and those with venous access issues.
Extravascular ICDs can be implanted safely and are able to detect and terminate induced ventricular arrhythmias.
Randomized trials comparing outcomes of subcutaneous ICDs with transvenous ICDs found device related complications and inappropriate shocks were similar in both groups.
Subcutaneous ICD have larger size, sure, the battery, life, and inability to provide pacing, including anti-tachycardia pacing to terminate ventricular arrhythmias.
The use of a single burst of ATP prior to shock in primary prevention ICD recipients can prolong the time to first all cause ICD shock (APPRAISE ATP investigators): the reduction in first all cause ICD shock did not extend to a reduction in all cause shock, burden or mortality as there was only a one percent absolute reduction in all cause shock due to ATP.
All current ICDs are MRI safe.
