 The immunoglobulin heavy chain (IgH) is the large polypeptide subunit of an antibody (immunoglobulin).
The immunoglobulin heavy chain (IgH) is the large polypeptide subunit of an antibody (immunoglobulin).
The IgH gene loci are on chromosome 14.
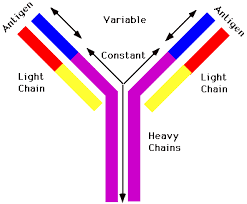
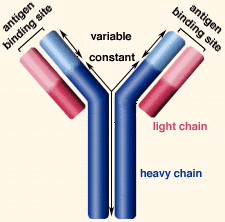
A typical antibody is composed of two immunoglobulin (Ig) heavy chains and two Ig light chains.
Several different types of heavy chain exist that define the class or isotype of an antibody.
All heavy chains contain a series of immunoglobulin domains, usually with one variable domain (VH) that is important for binding antigen and several constant domains (CH1, CH2, etc.).
Production of a viable heavy chain is a key step in B cell maturation.
If the heavy chain is able to bind to a surrogate light chain and move to the plasma membrane, then the developing B cell can begin producing its light chain.
The heavy chain doesn’t always have to bind to a light chain.
Pre-B lymphocytes can synthesize heavy chain in the absence of light chain, which then can allow the heavy chain to bind to a heavy-chain binding protein.
There are five types of mammalian immunoglobulin heavy chain: γ, δ, α, μ and ε: defining classes of immunoglobulins: IgG, IgD, IgA, IgM and IgE, respectively.
Heavy chains α and γ have approximately 450 amino acids.
Heavy chains μ and ε have approximately 550 amino acids.
Each heavy chain has two regions:
A constant region, which is the same for all immunoglobulins of the same class but differs between classes.
Heavy chains γ, α and δ have a constant region composed of three tandem immunoglobulin domains but also have a hinge region for added flexibility.
Heavy chains μ and ε have a constant region composed of four domains.
A variable region differs between different B cells, but is the same for all immunoglobulins produced by the same B cell or B cell clone.
The variable domain of any heavy chain is composed of a single immunoglobulin domain.
These domains are about 110 amino acids long.
