Only hematopoietic stem cells (HSCs) can regenerate HSCs and blood formation for life and transplant recipients, and HSCs represent only approximately one 100,000 of bone marrow cells.
Used for bone marrow transplant for a variety of hematologic disorders.
Identified by expression of CD34 and CD133.
Endothelial progenitor cells that reside in the bone marrow can promote neovascularization by secreting proangiogenic growth factors and can stimulate re-endothelialization and contribute to vascular homeostasis and possibly myogenesis.
HSCs circulate from bone marrow to blood to bone marrow, using homing receptors and chemokine receptors, and establish long lived hematopoiesis in other bones.
HSC aging may be accelerated by acquisition of somatic DNA mutations early in life.
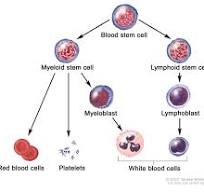
Hematopoietic stem cells (HSCs) are the stem cells that give rise to other blood cells (hematopoiesis).
Hematopoiesis occurs in the red bone marrow, in the core of most bones.
The red bone marrow is derived from the layer of the embryo called the mesoderm.
Hematopoiesis is the process by which all mature blood cells are produced.
The average person produces more than 500 billion blood cells every day and regulates the number of each blood cell type in the circulation.
The vast majority of hematopoiesis occurs in the bone marrow and is derived from a limited number of hematopoietic stem cells that are multipotent and capable of extensive self-renewal.
Hematopoietic stem cells give rise to different types of blood cells: myeloid and lymphoid.
Myeloid and lymphoid lineages both are involved in dendritic cell formation.
Myeloid cells include monocytes, macrophages, neutrophils, basophils, eosinophils, erythrocytes, and megakaryocytes to platelets.
Lymphoid cells include T cells, B cells, natural killer cells, and innate lymphoid cells.
Hematopoietic tissue contains cells with long-term and short-term regeneration capacities and committed multipotent, oligopotent, and unipotent progenitors.
Hematopoietic stem cells constitute 1:10,000 of cells in myeloid tissue.
HSC transplants are used in the treatment of cancers and other immune system disorders.
Hematopoietic stem cells resemble resemble lymphocytes,
are round, non-adherent, with a rounded nucleus and low cytoplasm-to-nucleus ratio.
The very first hematopoietic stem cells during embryonic development are found in aorta-gonad-mesonephros region and the vitelline and umbilical arteries.
Later, HSCs are also found in the placenta, yolk sac, embryonic head, and fetal liver.
Hematopoietic stem cells are found in the bone marrow of adults, especially in the pelvis, femur, and sternum. They are also found in umbilical cord blood and, in small numbers, in peripheral blood.
Stem and progenitor cells can be taken from the pelvis, as liquid to perform a smear to look at the cell morphology or they can be removed via a core biopsy, to maintain the architecture or relationship of the cells to each other and to the bone.
There are various kinds of HSC colony-forming units:
Colony-forming unit–granulocyte-erythrocyte-monocyte-megakaryocyte (CFU-GEMM)
Colony-forming unit–lymphocyte (CFU-L)
Colony-forming unit–erythrocyte (CFU-E)
Colony-forming unit–granulocyte-macrophage (CFU-GM)
Colony-forming unit–megakaryocyte (CFU-Meg)
Colony-forming unit–basophil (CFU-B)
Colony-forming unit–eosinophil (CFU-Eos)
Since hematopoietic stem cells cannot be isolated as a pure population.
IH is not possible to identify hematopoietic stem cells in a microscope.
Hematopoietic stem cells can be identified or isolated by the use of flow cytometry where the combination of several different cell surface markers, particularly CD34, are used to separate hematopoietic stem cells from the surrounding blood cells.
Hematopoietic stem cells lack expression of mature blood cell markers.
The lack of expression of lineage markers is used in combination with detection of several positive cell-surface markers to isolate hematopoietic stem cells.
Hematopoietic stem cells are characterized by their small size and low staining with vital dyes.
Hematopoietic stem cells can replenish all blood cell types, being multipotent and with the ability to self-renew.
A small number of Hematopoietic stem cells can expand to generate a very large number of daughter Hematopoietic stem cells, the phenomenon used in bone marrow transplantation.
In bone marrow transplantation a small number of Hematopoietic stem cells reconstitute the hematopoietic system.
Hematopoietic stem cells, like all adult stem cells, mostly exist in a state of quiescence, or growth arrest that is reversible.
The altered quiescent metabolism of HSCs helps the cells survive for extended periods of time in the hypoxic bone marrow environment.[
When triggered by cell death or damage, HSCs exit quiescence and begin actively dividing.
The transition from dormancy to propagation and back is regulated by the MEK/ERK and PI3K/AKT/mTOR pathways.
Dysregulation of these transitions can lead to stem cell exhaustion, or the gradual loss of active HSCs in the blood system.
Hematopoietic stem cells have a greater potential than other immature blood cells to pass the bone marrow barrier.
HSCs, may travel in the blood from the bone marrow in one bone to another bone.
HSCs that settle in the thymus, they may develop into T cells.
Hematopoietic stem cells may also settle in the liver or spleen and develop.
Hematopoietic stem cells to be harvested directly from the blood.
Hematopoietic stem cell transplantation (HSCT) is the transplantation of multipotent hematopoietic stem cells, usually derived from bone marrow, peripheral blood, or umbilical cord blood.
HSCs may be autologous or allogeneic.
HSCT is most often performed for patients with certain cancers of the blood or bone marrow, such as multiple myeloma or leukemia.[
The recipient’s immune system is usually destroyed with radiation or chemotherapy before the transplantation.
Infection and graft-versus-host disease are major complications of allogeneic HSCT.
To harvest stem cells from the circulating peripheral blood, donors are injected with a cytokine, such as granulocyte-colony stimulating factor (G-CSF).
G-CSF then induces cells to leave the bone marrow and circulate in the blood vessels.
Hematopoietic stem cell transplantation use has expanded beyond cancer to autoimmune diseases and hereditary skeletal dysplasias; notably malignant infantile osteoporosis and mucopolysaccharidosis.
Aging of hematopoietic stem cells are damaged by
DNA strand breaks that accumulate in long term hematopoietic stem cells.
This accumulation of DNA strand breaks is associated with attenuation of DNA repair and response pathways that depends on HSC quiescence.
The Non-homologous end joining (NHEJ) pathway repairs double-strand breaks in DNA, as the break ends are directly ligated without the need for a homologous template.
The NHEJ pathway depends on several proteins including ligase 4, DNA polymerase mu and NHEJ factor 1 (NHEJ1.
DNA ligase 4 (Lig4) has a highly specific role in the repair of double-strand breaks by NHEJ.
Deficiency of lig4 in pluripotent stem cells results in accumulation of DNA double-strand breaks and enhanced apoptosis.
NHEJ is a key determinant of the ability of stem cells to maintain themselves against physiological stress over time.
DNA damage accrual may be an important physiological mechanism of stem cell aging.
The clonal diversity of hematopoietic stem cells is greatly reduced around age 70.
