 More than 50,000 cases of DCIS will be diagnosed in 2022 in the US.
More than 50,000 cases of DCIS will be diagnosed in 2022 in the US.
Highest risk is associated with heterogeneous fine pleomorphic morphology with the clustered group distribution, as compared with the coarse calcifications, which include a low likelihood of DCIS.
Fine linear branching microcalcifications on mammogram is associated with a higher rate of local recurrence.
Digital mammography is more sensitive for DCIS detection compared to film screen mammography.
Much of the burden of overdiagnosis on mammography is a result of detection of ductal carcinoma, in situ, a diagnosis faced by more than 50,000 women in the US annually.
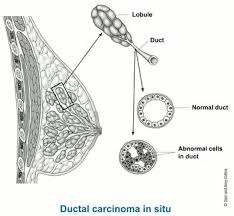
Characterized by proliferation of neoplastic epithelial cells within the lumen of the breast, with an intact basement membrane.
Most commonly presents is microcalcifications in the absence of other clinical findings.
Encompasses a heterogenous group of lesions with variable history and risk to progress to invasive breast cancer.
A non-obligate precursor lesion for infiltrating ductal carcinoma.
Recommended evaluation for DCIS includes bilateral diagnostic mammography, pathology review, determination of tumor estrogen receptor status, and MRI, as indicated.
Post treatment survival is approximately 98%.
Up to 50% of DCIS lesions progress to invasion following diagnostic excision alone.
After wideexcision invasive recurrences are halved by radiotherapy use.
Radiotherapy however, is not needed for all patients: low intermediate grade DCIS, lesions 2.5 cm or less, and high grade DCIS of 1 cm or less.
ER status should be assessed to determine the benefit of adjuvant endocrine therapy or risk reduction.
HER2 status prognostic importance in DCIS has not been concluded.
Knowing HER2 status in DCIS does not alter its management.
328% increase in age-adjusted incidence in the past 10 years.
Prior to mammography use DCIS was a relatively uncommon finding.
The majority of cases are diagnosed as asymptomatic microcalcifications on screening mammography, with 80% presenting as microcalcifications alone and 20% as other suspicious mammographic findings, including masses and focal nodular patterns.
Accounts for about 20%-25% of all female breast cancers.
Diagnosed 10 times more frequently than before the use of screening mammography.
Only 5% of cases are associated with the palpable finding.
It has an exceptionally low mortality of 3% at 20 years, and there is no demonstrable survival benefit from the addition of breast radiation, endocrine therapy, or a combination of these treatments to lumpectomy.
MRI has a sensitivity of up to 98% for diagnosis high-grade DCIS, but overall does not improve management or results in DCIS.
One fourth as common as invasive breast cancer, with an age-adjusted incidence 32.5 cases per 100,000 women in the US per year.
More than 60,000 cases diagnosed annually.
Approximately 500,000 women in the United States or living with DCIS.
A premalignant process, with the rate of development of invasive carcinoma 14-60% with a followup period of up to 30 years.
Traditionally considered a precursor to invasive breast cancer, however incidence and treatment of DCIS has risen, and there has not been a concomitant decline in the incidence of breast cancer.
DCIS detection has increased over recent decades secondary to mammographic improvements, advanced stage distributions have not correspondingly declined, suggesting that screening may detect some DCIS that would remain subclinical.
Women who are diagnosed with ductal carcinoma in situ (DCIS) on screening mammography are more than twice as likely to develop invasive breast cancer and die from it than women who do not have DCIS, even those with low and intermediate grade DCIS, a population-based, observational cohort study has shown.
In a small number of untreated patients, death rate from breast cancer was 10-20%.
Relative survival is extremely high, greater than 98%, with or without radiation or anti-estrogen therapies.
Presently, with treatment there is a 3.3% death rate for patients with DCIS
Presently it is viewed as a non-obligate precursor to invasive disease.
Progression to invasive disease is not definite, and the process of transitioning to invasive breast cancer is likely to be the result of biologic interplay between liquidation of genetic events and a permissive microenvironment.
A heterogeneous process, with an excellent prognosis when treated.
A non-obligate precursor of invasive ductal carcinoma, although incompletely excised DCIS does not recur or become life-threatening in the majority of cases.
The risk of breast cancer mortality is elevated in patients with DCIS compared with the general population, with risk being higher among younger versus older women and black versus white women.
Approximately 50% of breast cancer deaths occur without development of a breast invasive recurrence.
Nonpalpable mammographically detected DCIS has a better prognosis than palpable DCIS, which is often associated with microinvasion and occasionally with axillary nodal involvement.
Accurate preoperative imaging is required to determine the size and extent of the disease to optimize surgical management.
Patients with microinvasion have a prognosis almost identical to women with DCIS alone.
Primary therapy is to prevent progression to invasive breast cancer.
7-20% of patients with DCIS thought to be at low risk, are actually found to have invasive carcinoma when a core-needle biopsy shows DCIS, and there will be a potential delay in diagnosis of invasive disease in the absence of lumpectomy.
Management includes: surgery-mastectomy or lumpectomy with or without radiation therapy, and adjuvant endocrine therapy to reduce risk of recurrence.
The goal of primary therapy for DCIS is to prevent progression to invasive breast cancer.
Management strategies for DCIS include surgery (mastectomy or breast conserving surgery), radiation therapy, followed by adjuvant endocrine therapy in eligible patients to reduce risk of recurrence.
The choice of local treatment does not impact overall survival and the patients individual preference and risk of local recurrence.must be considered.
Sentinel lymph node biopsy should be considered at the time of definitive surgery to avoid necessitating a full axillary lymph node dissection.
RTOG 9804 trial at 7 years showed the local recurrence rate after lumpectomy and whole breast radiation was associated with a local recurrence rate of 0.9% and 6.7% in the lumpectomy only arm: there is no change in distant metastasis free survival.
In higher risk DCIS-higher nuclear grade, younger age, large tumor size there may be a statistically significant improvement in overall survival with whole breast radiation therapy: there is a suggestion of a small but significant reduction in breast cancer mortality.
The use of a radiation therapy boost provides a small but statistically significant reduction in ipsilateral breast tumor recurrence at 4% at 20 years for invasive breast cancers.
Meta analyses of multicenter trials demonstrate that the addition of whole breast radiation after lumpectomy provides a statistical and clinically significant reduction in ipsilateral breast events, distant metastases free survival. but no overall survival benefits.
Patient who have previously received radiation therapy for DCIS have higher mortality when developing an invasive second breast cancer than those who did not receive radiation.
The choice of local treatment does not impact overall disease associated survival.
Large randomized trials demonstrate the addition of whole breast radiation after breast conserving surgery for DCIS provides a statistically and clinically significant reduction in ipsilateral breast cancer events, but does not show the addition of RT has an overall survival benefit.
NSABP 15 year results showed a 52% reduction of ipsilateral invasive recurrence invasive with radiation after excision versus excision alone.
10 year local recurrence rate after breast conserving surgery and RT for patients with negative margins of <2mm vs >2mm is not significantly different, both being less than 5%.
Wider margins are significantly associated with the lower rate of recurrence only in patients who do not receive radiation, but not in those treated with radiation.
The use of at least a 2 mm margin in DCIS treated with RT is associated with low rates of ipsilateral breast cancer recurrence.
Prognosis favorable and diagnosis of DCIS not likely to affect a woman’s survival.
The risk of breast cancer mortality 10 years after the treatment of DCIS approximates 4%.
Women younger than 40 of age have a recurrence rate 7 times women older than 80 years, and twice that of women older than 50 years.
An observational study of more than 100,00 patients with DCIS had a mortality of brest cancer of 3.3% (Narod et al).
The COMET study found that women with low risk DCIS randomized to active monitoring do not have a higher rate of invasive cancer in the same breast at two years compared with those randomized to guideline concordant care.
The COMET trial provides information in patients with DCS and a limited life expectancy, surgical therapy and possibly even endocrine therapy can be omitted.
The long-term risk of invasive cancer development with active monitoring of patients with low grade DCIS in older women is unknown, but appears to be at least 0.6% per year in the absence of surgery.
DCIS is more lethal when it occurs in women younger than 35 years of age and in black women.
Mammograms detect calcifications and residue in ducts from necrosis but does not identify the neoplasm size accurately.
Bulky, palpable disease is rare.
Patients with involvement of 2 or more quadrants should have a total mastectomy without lymph node dissection.
For patients undergoing mastectomy for DCIS a sentinel lymph node biopsy procedure is strongly considered at the time of surgery.
Mastectomy is curative in 95-100% of patients.
In the ECOG 5194 study of the natural history of DCIS treated with lumpectomy alone: 565 women with low or intermediate grade disease between 0.3-2.5 cm in diameter or 105 women with high grade disease between 0.3-1 cm in diameter (approximately 30% of women in each group took tamoxifen for some time)-after a median followup of 6 years, the five-year rate of invasive cancer in the same breast was 6.1% in the low-grade group and 15.3% in the high-grade group, while in the opposite breast to rates with 3.7% and 7.4% respectively.
Stratification of risk based on size, grade, presence of comedo necrosis, margins, and patient age.
Local recurrence rate after mastectomy <5%.
20-25% of all new screen detected breast cancer cases.
Accounts for one in five neoplasms detected on mammography.
Rate of detection has increased from 1973 to 1972 with an increased incidence of 557% compared to a 34% increase in the diagnosis of invasive breast cancer.
Over 90% diagnosed by mammographic examination alone.
Mastectomy and breast conserving surgery with and without radiation provide equivalent outcomes and treatment selected on the basis of patient’s wishes and clinicopathologic factors.
Selected patients with a low risk of breast recurrence when treated with excision alone have a 94% 10 year disease risk free survival and 83% for patients with intermediate and high-risk the DCIS.
Risk stratification is based on tumor size, grade, absence of comedonecrosis, margin width , and age at diagnosis.
Lumpectomy is now the primary treatment for most patients, although in recent years rate of mastectomy increasing.
Whole breast irradiation addition to a margin free excised pure DCIS lesion decreases the rate of in-breast disease recurrence by approximately 50%, but does not affect overall survival, or distant metastasis free survival.
Low rates of local recurrence in patients with DCIS can be achieved with radiotherapy, however invasive recurrences can represent more than 50% of all local recurrences and such occurences are associated with higher mortality rates.
Studies suggest a substantial and ongoing risk of local recurrence with excision alone in patients with DCIS.
SEER study Of 108,196 women diagnosed with DCIS: A 20 years overall breast cancer specific mortality was 3.3% with the risk being 1.8 times higher versus the general population.
For DCIS lesions that are not considered high risk, radiation therapy should not be given routinely after lumpectomy because it does not affect mortality.
Trials have shown disease free survival 94% in patients with low grade DCIS without RT, and 83% for patients with intermediate and high-risk DCIS.
The use of a radiation boost to the tumor bed is recommended to maximize local control, especially in patients with DCIS under 50 years of age.
Gene expression assays can be used to decide if radiotherapy is necessary to reduce the risk of recurrence, and the avoidance of radiation therapy.
In DCIS patients treated with lumpectomy and whole breast radiation, with boost RT or no boost: a decrease in tumor recurrence was seen in the boost group at 15 years of 91.65 vs 88% in a pooled analysis.
Boost radiation has negative influence on patient reported cosmetic outcomes. that is clinically meaningful.
P53 mutations seen in up to 25% of cases of DCIS.
Increased risk of local relapse in patients with p53 overexpression.
In lesions 2 cm or less in diameter, the risk of ipsilateral breast event, either recurrent disease or invasive cancer is 28.1% at 10 years and can be reduced to 12.9% at 10 years with adjuvant radiation: Adjuvant radiation does not influence overall survival.
Associated with approximately a 10% risk of invasive ipsilateral breast tumor recurrence, even in the treated population without radiation.
In a study of women treated for this entity in Ontario Canada from 1994-2003, for women aged 50 years and younger than her recurrent cancer rate was nearly twice as high for women aged 45 years or younger:in this retrospective study 17% of women experience local recurrence, 6.5% or invasive lesions, women under the age of 45 years had an approximately 22% rate of recurrence in women greater than 50 years a 10-15% chance of recurrence within 10 years of diagnosis (Kong I).
Randomized clinical controlled trials have demonstrated that adding radiation after surgical excision for DCIS reduces the risk of developing local recurrence and invasive local recurrence by approximately 50%.
Most patients will not develop local recurrence if treated using surgical excision alone, in DCIS without radiation, and many patients are treated in contemporary practice using surgical excision alone.
Using the Oncotype DX score can quantify ipsilateral breast event risk and invasive breast event risk and can improved selection of patient treatments for the DCIS: For DCIS risk groups of low, intermediate, and high risk, the 10 year risk of development of an ipsilateral breast event or 10.6%, 26.7%, and 25.9% respectively, and for invasive ipsilateral breast events 3.7%, 12.3%, and 19.2% respectively (Solon LJ et al).
In the above study the population selected was treated with surgical excision alone without radiation, and the risk of an ipsilateral breast event was more than twofold higher for the 30% of patients with an intermediate or high DCIS score compared with the 70% of patients with a low DCIS score.
Women with diagnosed DCIS or LCIS have an approximately 4 times risk of developing invasive breast cancer than the general population (Warnberg).
Up to 20% of women with DCIS who have a recurrence within 5 years, and half of those will be invasive in type.
Approximately 50% of local recurrences following breast consrving treatment will be invasive cancers and 50% will be DCIS.
Compared to women without prior DCIS women with DCIS 2-4 fold higher risk of developing cancer in the contralateral breast (Habel LA).
Fewer than 6% of women with DCIS, develop contralateral breast cancer within 10 years.
Two large randomized studies evaluating the addition of tamoxifen to postoperative radiation following lumpectomy showed conflicting results.
In a recent study there was no statistical difference in local recurrence rates between patients with negative margins on initial resection than those with negative margins after reexcision indicating that getting negative margins is vital to maintain local control.
National Surgical Adjuvant Breast and Bowel Project B-24 reported that women with ER+ DCIS who received tamoxifen after lumpectomy and radiation had a 40-50% reduction in subsequent breast cancer at 10 years compared with women who received placebo.
NSABP B-24 trial, 1804 women with DCIS randomly assigned to receive five years of adjuvant tamoxifen or placebo following breast conserving therapy and whole breast radiation: at 83 months follow up women in the tamoxifen group had fewer breast cancer events then those in the placebo group, 10.3% versus 16.9%.
In the above study the cumulative incidence of all breast cancers in the tamoxifen group was 4.8% at seven years, 2.6% in the ipsilateral breast and 1.8% in the contralateral breast and 0.4% in regional and distant sites.
National Surgical Adjuvant Breast and Bowel Project B-35 (NSABP-35) tamoxifen compared to a anastrozole treatment in postmenopausal women with DCIS undergoing a lumpectomy plus radiotherapy: anastrozole treatment provided significant improvement in breast cancer free interval mainly in women younger than 60 years of age.
No reliable predictors of residual disease in reexcision specimens from patients with DCIS.
Margins >10 mm may be excessive and lead to less optimal cosmetic result.
Margin <1mm is considered inadequate.
With pathologic margins between 1 and 10 mm , wider margins are generally associated with lower local recurrence rates.
The guiding principles for reexcision are margin status and postoperative mammogram residual calcifications.
Postexcision mammography is valuable to confirm that an adequate excision of DCIS has been performed, particularly for patients with initial presentation of microcalcifications.
Treatment with excision and radiation therapy associated with recurrence rates ranging from 7-19% and approximately 50% of recurrences are invasive.
No clinical relevance when micrometastatic foci of disease or isolated tumor cells are found in axillary lymph nodes.
All women with locally excised ER+ ductal Ca in situ should be offered adjuvant endocrine therapy to include tamoxifen or aromatase inhibitors depending upon menopausal status.
It is difficult to prove in postmenopausal women with DCIS and ER positive status that tamoxifen or aromatase inhibitors are noninferior, because of the excellent prognosis of the underworld process..
Cumulative 5-10 year probabilities of being diagnosed with a contralateral breast cancer among women diagnosed with DCIS is 4.3% and 6.8%, respectively.
In a retrospective study 16.8% of patients with DCIS were diagnosed with an unrelated cancer before or after the diagnosis of breast cancer.
Approximately 60% of patients will progress to invasive cancer within 8-10 years.
Most studies have demonstrated to have an association between nuclear grade and or tumor necrosis and the risk of ipsilateral breast tumor recurrence.
In a double-blind study randomized placebo-controlled trial women diagnosed with hormonally receptive positive DCIS were randomized to 1 mg anastrozole or 20 mg of tamoxifen for five years: no clear efficacy differences were seen between the two groups in preventing locoregional and contralateral breast cancer in postmenopausal women (Forbes JF et al).
Van Nuys Prognostic Index (VNPI) by Silverstein and others stratifies patients with DCIS into low, intermediate and high-risk groups based on tumor grade, size and margin width.
VNPI scores of 3-4, low risk-treatment recommended excision alone, score 5-7, intermediate risk-treatment recommended excision with radiotherapy and patients with score of 8-9 recommended to have mastectomy.
National Surgical Adjuvant Breast and Bowel Project (NSABP) B17-enrolled 818 patients and compared lumpectomy alone with lumpectomy and whole breast irradiation-median follow-up more than 10 years, breast irradiation was associated with a reduction in the rate of invasive ipsilateral breast tumor recurrence from 17% to 8% and that of all noninvasive ipsilateral breast tumor recurrence was reduced from 15% to 8%.
The NSABP B-17 trial showed at 15 years the radiation arm had a 52% reduction of ispilateral invasive recurrence compared with excision alone, without an alteration in survival data.
National Surgical Adjuvant Breast and Bowel Project (NSABP) B24 randomized 1804 patients with DCIS 1 cm or smaller, treated with radiation and tamoxifen or placebo: Recurrent or invasive cancer was reduced by 37% with tamoxifen from 13.4% to 8.2% at 5 years, the overall survival is 97% and not significantly different between the groups.
European Organization for Research and Treatment of Cancer (EORTC) 10853 trial found that irradiation after lumpectomy associated with a reduction in both invasive and DCIS tumor recurrence of approximately 50%.
The present recommendation guidelines include breast conserving surgery plus radiation, which can mean overtreatment of many women with DCIS of 2 cm or less, given that wide excision alone results in effective cancer control of the breast in more than 70% of cases at 10 years.
Current DCIS protocols may constitute gross overtreatment, exposing thousands of women to therapy-related morbidity unnecessarily.
A phase 3 NSABP B-35 trial comparing anastrazole 1 mg daily versus tamoxifen 20 mg daily for 5 years in postmenopausal women with ER receptor or PR receptor positive ductal carcinoma in situ-3104 patient’s randomized and after a median follow-up of 8.6 years the estimated 10 year breast cancer free interval was 89.2% in the tamoxifen group and 93.5% in the anastrazole group, with significantly fewer cases of invasive breast cancer in the anastrazole group.
In the above study contralateral breast cancer was reduced by almost half.
Anastrozole provides at least comparable benefit as agent treatment for postmenopausal patients with hormone receptor positive DCIS treated with breast conserving surgery and RT with a different toxicity profile.
Breast conserving surgery without radiation is a reasonable option when risk appears to be low.
In a population based study, the use of whole breast radiation in patients with high-risk DCIS with higher nuclear grade, younger age, and larger tumor size have a modest improvement in survival (Sagara Y).
It is not possible to accurately identify which patients are at highest risk for recurrence of DCIS or developing invasive cancer using clinical pathological features alone.
The majority of recurrences of DCIS are in-breast recurrences after breast conserving therapy and recurrences mostly occur in close proximity to the location of the prior disease.
Overall, approximately 1/2 of local recurrences after initial treatment of a pure DCIS are invasive in nature, whereas the remainder occur as pure DCIS.
Breast cancer mortality is not reduced with prevention of ipsilateral invasive recurrence by the addition of radiotherapy to lumpectomy or with unilateral mastectomy versus lumpectomy.
Patients with ER positive DCIS treated with BCT recommended to have endocrine therapy with tamoxifen or an aromatase inhibitor to reduce the risk of ipsilateral breast cancer recurrence.
In a phase 3 study anastrozole was found to be a better option than tamoxifen as an adjuvant treatment in postmenopausal women with DCIS: patients who received anastrozole will more likely to be free of breast cancer at 10 years after randomization 93.5% versus 89.2%.
The above study tamoxifen had a higher rate of uterine cancers and a lower rate of osteoporotic fractures and patients with tamoxifen will more likely to have a thromboembolic event.
DCIS score, also known as the Oncotype DX Breast Cancer Assay for DCIS uses seven genes predictive of recurrence risk and 5 references genes.
DCIS score calculation scaled from 0-100.
DCIS score risk categories: Less than 39, low risk, 39-54, intermediate risk, 55-100, high risk.
Mastectomy management is performed more often with DCIS than for invasive cancer.
In a population-based retrospective longitudinal cohort study from SEER involving
57,222 cases of DCIS the survival benefit of performing breast surgery for low-grade DCIS was lower than for intermediate or high-grade DCIS (FallowfieldL et al).
Among 35,024 women who were diagnosed with DCIS through the NHS invasive breast cancer developed in 8.82 per 1000 women per year and did not vary significantly with age at diagnosis,
This rate was more than double the expected rate of breast cancer incidence in the general population
The rate of death from breast cancer was 1.26 per 1000 women per year which was 70% higher than that expected from national breast cancer mortality rates at an observed to expected ratio of 1.70.
The death rate did not vary significantly with age of DCIS diagnosis.
For both invasive breast cancer and death from it, the increased risks continued for at least 20 years.
13,606 women had been followed for up to 5 years; 10,998 for 5 to 9 years; 6,861 for 10 to 14 years; 2,620 for 15 to 19 years, and 939 for at least 20 years.
2076 women in the study had developed invasive breast cancer.
Beyond three years, the observed to expected ratio was 2.69 with no further increase over time.
After 5 years, the breast cancer death rate increased with values of 1.98 in years 5 to 9; 2.99 in years 10 to 14, and 2.77 15 years and more after the diagnosis of DCIS.
Women treated more aggressively had lower rates of developing ipsilateral invasive breast cancer over time.
The rate of ipsilateral invasive breast cancer among women who underwent breast conserving surgery, but no radiotherapy was 43% higher at an adjusted rate ratio of 1.43 compared to women who had breast-conserving surgery accompanied by radiation.
For women who underwent mastectomy, the rate of ipsilateral breast cancer was 35% lower at an adjusted rate ratio of 0.65 again compared to women who had breast-conserving surgery accompanied by radiation.
Furthermore, the cumulative rate of invasive breast cancer increased more steeply for women who had undergone breast conserving surgery—with or without radiotherapy—than for those who had undergone a mastectomy.
The adjusted rate ratio for women with involved margins was 3.73 compared to women whose final margin distance was at least 5 mm.
Moreover, the rate of ipsilateral invasive breast cancer among women with estrogen receptor-positive disease treated with endocrine therapy was 38% lower.
Fifteen years after the diagnosis, the cumulative rate of ipsilateral invasive breast cancer was 4.7% in women with estrogen receptor-positive breast cancer treated with endocrine therapy compared with 7.3% in women who were not treated with hormonal therapy.
The 15-year cumulative rate of ipsilateral invasive breast cancer among women with low and intermediate grade DCIS was higher than that of women with high grade tumors.
Women diagnosed as having DCIS detected by screening
have experienced substantially increased risk of both invasive breast cancer and death from breast cancer compared with women in the general population, despite having lower overall mortality.
Death rates from breast cancer among women with a more recent diagnosis of DCIS are declining, likely a reflection of improved treatment of invasive disease.
DCIS diagnosis on screening mammography more than doubles the risk of invasive breast cancer and death over a long-term follow-up even among those with low and intermediate grade DCIS.
