 Neurostimulation therapy includes invasive and noninvasive approaches that apply electromagnetic energy to specific anatomical targets to ninduce neuromodulation of the corresponding neural circuitry.
Neurostimulation therapy includes invasive and noninvasive approaches that apply electromagnetic energy to specific anatomical targets to ninduce neuromodulation of the corresponding neural circuitry.
Invasive neurostimulation are effective treatments for a growing number of medically resistant neurologic and neuropsychiatric disorders.
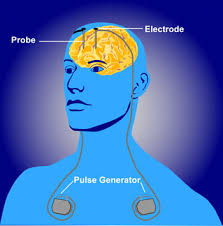
Wires are placed into the brain and pulses of electricity travel from a battery to the electrodes at the tip of the wires.
Deep brain stimulation requires intracranial placement of thin leads on one side, or both, sides of the brain, typically into the subthalamic nucleus or globus pallidus, connected to a neurostimulator subcutaneously in the subclavicular region.
The scalp is anesthetized locally and a burr hole is placed in the skull.
The surgical procedure is generally performed while the patient is awake and utilizes a stereotactic localizing system.
Mid line anatomical structures provide landmarks for target planning.
Magnetic resonance imaging, CT scanning, stereotactic atlases and micro electrode recordings identify the deep nuclei.
Microelectrode recordings allow for stimulation of the target area and also help in the placement of the electrodes.
More than 100,000 cases have been performed.
Local anesthesia is provided to the scalp and a bur hole is made in the skull, and identification of the deep nuclei and is made with MRI or CT scans, stereotactic atlases and microelectrode recordings.
Microelectrode recordings allow for targeted area stimulation and aid in the placement of permanent electrodes.
Following electrodes placement, lead extensions and an impulse generator are surgically implanted.
Most often the battery source for the neurostimulator is placed in the sub clavicular region.
The device provides programming with therapeutic options and has the ability to have ongoing adjustments.
DBS acts on the cells and fibers located closest to the implanted electrodes and in most cases inhibit cells and excites fibers.
The normal human brain functions through rhythmic oscillations that continuously repeat, and when there is change in these oscillations they can affect cognitive, behavioral, and motor function.
As a result alterations it in rhythmic oscillations can result in disabling tremor or other symptoms of Parkinson’s disease.
Deep brain stimulation can be performed in both cerebral hemispheres to control bilateral symptoms.
DBS influences multiple thalamocortical circuits, downstream pathways and other CNS structures.
Changes the firing rate and pattern of individual neurons in the basal ganglia, and the electrical current acts at synapses and triggers neighboring astrocytes to release calcium and promote local release of neurotransmitters such as adenosine and glutamate.
DBS increases blood flow and stimulates neurogenesis.
The effects of DBS occur across a large neural network, extending beyond local neuronal cell bodies and axons around the electrical field.
D S has become the main surgical treatment for patients with Parkinson’s disease with fluctuating response to levodopa.
Can induce analgesia and provide intractable neuropathic and nociceptive pain.
DBS is an open surgical procedure and carries a modest risk of intracranial bleeding and infection, and some patients find it to be intrusive and unacceptable.
DBS targets include sensory nuclei of the thalamus, peraqueductal/periventricular gray, anterior cingulate cortex, internal capsule, posterior hypothalamus, and nucleus accumbens.
Deep brain stimulation of the nucleus accumbens was successful in decreasing depression symptoms in 50% of patients who did not respond to other treatments such as electroconvulsive therapy.
Risks of deep brain stimulation are transient confusion, hemorrhage, infection, lead fracture, misplacement or migration.
Infection is the most commonly reported complication.
Morbidity rate of deep brain surgery is 3-4% (Voges J et al).
Techniques utilized for movement disorders such as Parkinson’s dystonia, and tremor.
Patients with Parkinson’s disease without significant cognitive or psychiatric problems and have motor fluctuations, intractable trauma, or intolerance of medications are good candidates for DBS.
DBS improves levodopa responsive symptoms, dyskinesia and tremors.
Subthalamic DBS may increase depression, apathy impulsivity, and impair verbal fluency.
May be used for chronic pain associated with amputation, stroke anesthesia dolorosa, and head pain.
Adverse risks include intracranial bleeding, lead dislocation, lead fracture, and infection.
Can lead to stimulation induced adverse effects such as aggression, depression, mania, prolonged erection and inappropriate laughter.
Electrical brain stimulation changes brain activity in a controlled and reversible manner.
Mode of action is stimulation induced brain activity modulation.
Surgical intervention of choice for Parkinson’s disease when motor complications are inadequately controlled with medications.
Best candidates for this procedure in Parkinson’s disease are patients with fluctuating motor symptoms, suffering with adverse medication effects, have few comorbidities, no behavioral or cognitive problems.
Utilized in patients with a high likelihood that their lives will be improved and when all other interventions have been tried.
Emerging treatment for Parkinson’s disease, dystonia depression and a excessive compulsive disorders.
