1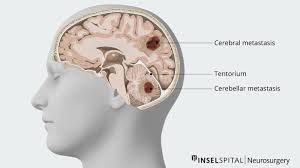
Estimated 200-250,000 newly diagnosed cases occur annually in the U.S., comparable to the incidence of primary breast, prostate, lung, and colorectal cancer.
Metastatic brain tumors are the most common intracranial neoplasms in adults.
Unlike lung, liver or bone metastasis brain metastasis are generally less responsive to systemic therapy, such as chemotherapy, because the blood brain barrier impedes drug penetration:consequently, radiation therapy is commonly used.
Two radiation modalities are used to treat brain metastasis; whole brain radiation delivering a moderate dose of radiation to the entire brain, including tumors and normal tissue, and stereotactic radiation, focusing only on visible tumors with higher biological doses.
Stereotactic radiation preserves neurocognitive function, and patient reported outcomes compared with whole brain radiation.
Stereotactic radiation improves symptoms and interference, key components of quality of life, compared with hippocampal avoidance whole brain radiation in patients with up to 20 brain metastases.
Patients with NSCLC have a higher prevalence of ALK fusions and EGFR mutations in patients who eventually develop brain metastasis, due to their longer survival.
Leptomeningeal metastases occur in 3 to 4% of patients with non-small cell lung cancer.
Morbidity associated with brain metastasis is a serious concern, as quality-of-life is markedly reduced among patients with brain tumors.
Estimated incidence is high as 10 per 100,000 thousand population and over 100,000 patients are expected to develop brain metastases each year.
Autopsy studies estimate the incidence of CNS involvement at 40-75% of the time of death depending on the primary Cancer type.
It accounts for approximately 140,000 cases of cancer mortality annually in the United States
Common clinical manifestations include: headache, neurologic defects, cranial nerve, motor and sensory deficits, cognitive impairment, and seizure activity.
A population based study in Sweden of payients admitted to hospitals revealed a doubling of annual age adjusted incidence rate of hospitalization for brain metastases from 7-14 patients per 100,000 population between 1987 and 2006 (Smedby KE).
The rising incidence of intracranial brain metastases over the past two decades is attributed to increased detection with MRI imaging, and, paradoxically, improved systemic disease control.
Brain metastasis are usually multiple lesions spread at the gray-white junction and/or smooth margins of the brain.
Small brain tumors may present with or without vasogenic edema.
Interplay between cell surface receptors, neurotropic growth factors, and brain vasculature play a role in development and site of brain metastases.
Most common intracranial tumors in adults.
Estimated incidence to be 10 times more common than primary brain tumors.
Most common complication of systemic malignancy.
Brain metastases are more common in non-small cell lung cancer adenocarcinoma type than in patients with squamous histology.
5 to 10% of patients with NSCLC have concurrent brain metastasis at the time of diagnosis, and 20 to 40% will eventually develop brain metastases.
Of patients presenting with metastatic disease, melanoma is most likely to present with synchrous brain metastases, followed by adenocarcinoma of the lung, non-small cell lung cancer, small cell lung cancer, bronchoalveolar carcinoma, squamous cell lung cancer, and renal cancer.
As many as 40 to 60% of patients with small cell lung cancer develop brain metastasis during the course of their disease.
Patients with small cell lung cancer show a 1.3 to 2 times higher risk of developing brain metastasis in patients with non-small cell lung cancer, and a reduced time to intracranial disease progression.
Occurs as a result of hematogenous metastases.
100,00 people die per year with symptomatic intracranial metastases.
Lifetime risk of brain metastases in cancer patients is 15-30%.
Most common primary tumors associated with brain metastases are lung (40%), breast (19%), unknown primary (10-15%, prostate (10%) and colon (5%) cancer.
Breast cancer is the second most common cause of brain metastasis.
Melanoma is the solid tumor with the highest risk for dissemination to the CNS
Melanoma brain metastases incidence at autopsy is up to 70%.
Autopsy studies indicate 15-35% of patients with breast cancer found to have brain metastases, not all of which are clinically apparent prior to death.
The risk of brain metastases in breast cancer is subtype dependent, and patients with human epidermal growth factor receptor 2 positive and triple negative subtypes experience significantly higher rates of CNS relapse than patients with hormone receptor positive and HER2 negative tumors.
In advanced HER2 positive breast cancer up to 30% of patients develop brain metastasis, and in triple negative breast cancer incidence of brain metastasis with advanced disease Iis as high as 50%.
EBBR2 positive breast cancer‘s have a higher risk of intracranial metastases then patients with hormone receptor positive/ERBB2 negative disease.
Multiple metastases commonly associated with melanoma and lung primaries while single lesions more common with breast, colon and renal cell cancers.
Headache is usually mild at onset, begins on awakening, disappears shortly afterwards, and returns the following morning, but is present only in 1/4-1/3 of patients.
Approximately 20% of patients with metastatic melanoma have brain metastases at diagnosis.
Long-Term Survival of Patients With Melanoma With Active Brain Metastases Treated With Pembrolizumab on a Phase II Trial
Melanoma is the solid tumor with the highest risk for dissemination to the CNS
Long-term follow-up of pembrolizumab-treated patients with new or progressing brain metastases treated on a phase II clinical trial.
A study that enrolled 23 patients with melanoma with one or more asymptomatic, untreated 5- to 20-mm brain metastasis not requiring corticosteroids; 70% of patients had prior systemic therapy.
Pembrolizumab was administered for up to 24 months.
Six patients (26%) had a brain metastasis response.
The median progression-free and overall survival times were 2 and 17 months, respectively.
48% were alive at 24 months, suggesting pembrolizumab is active in melanoma brain metastases with acceptable toxicity and durable responses.
Melanoma brain metastases incidence at autopsy is up to 70%.
Whole-brain radiation is typically ineffective for melanoma.
All patients with extracerebral response also responded in the CNS.
The median PFS time was 2 months, and the median OS time was 17 months.
At 24 months of follow-up, 11 patients (48%) remained alive
23 patients were enrolled and although the brain metastasis RR was only 26%, three patients who were unevaluable for CNS response remained alive and progression free at 24 months.
All patients who responded systemically also responded in the CNS, and all responses were ongoing for the 24-month duration of the study.
The median OS time was 17 months, and 48% of patients were alive at 2 years.
Brain metastasis response rates are greater than 50% in BRAF-mutant melanoma, treated with BRAF/MEK inhibitors, although the duration of response tends to be short.
Ipilimumab, administered with untreated brain metastases, was less active than PD-1 inhibitors, similar to extracerebral disease.
PD-1 inhibitors have activity in patients with NSCLC and brain metastases with at least 1% PD expression.
In a trial of nivolumab or nivolumab plus ipilimumab in patients with brain metastasis; the RRs to monotherapy and combination therapy were 20% and 42%, respectively, in asymptomatic patients.
Patients previously treated with BRAF/MEK inhibitors had a worse outcome.
Tawbi presented early results of a trial of ipilimumab and nivolumab in patients with untreated with brain metastasis, demonstrating a brain metastasis RR of 55%.
Combination therapy should be considered for patients with brain metastases.
All patients who achieved an objective response remained in response at 24 months, which is longer than the CNS response in patients treated with BRAF/MEK inhibitors.
Brain metastases, particularly when small and not located in critical sites can safely be treated with systemic therapy or systemic therapy combined with local therapy.
Prophylactic antiepileptics are recommend for patients with untreated brain metastases treated with immune therapy.
The incidence of radionecrosis (30.4%) was higher than expected based on historical observations, which may be as a result of immune therapy, improved longevity, or both.
There is no clear threshold for PD-L1 positivity below which responses are not seen.
Cerebral hemispheres are the site of 80% of melanoma brain metastases, followed by cerebellum (15%) and brainstem (5%).
Preoperative stereotactic radiosurgery reduces cavity, local recurrence, meningeal disease, and adverse radiation rates in patients with resectable brain metastasis.
Stereotactic radiosurgery for brain metastasis results in local control rates of 50-75% at one year.
Stereotactic radiosurgery has a sustained tumor control rate of 73%.
Stereotactic radiosurgery of the surgical cavity in patients who have had complete resection of one, two, or three brain metastases significantly lowers local recurrence compared to observation alone, and the use of SRS after brain metastasis resection could be an alternative to whole-brain radiotherapy (Mahayana A).
Survival outcomes or equitable for treatment with SRS compared with whole brain radiation in patients with small cell lung cancer and brain metastasis.
Oligometastatic disease refers to metastatic disease limited to 5 or fewer lesions.
Autopsy studies of breast cancer patients reported to have brain metastases in 6.7%-36% of cases.
Capecitabine has efficacy for breast cancer brain metastases (Ekenel M).
Incidence increasing and has a median survival of 5-11 months.
Patients with a single metastases survive longer with WBRT plus STS than with WBRT alone.
STS is reserved for limited volume brain metastases and WBRT for more widespread brain metastases.
In a randomized clinical trial patients with resected, limited number of brain metastasis, the use of SRS alone compared with WBRT resulted in less cognitive deterioration among long-term survivors: Both local and distant intracranial tumor controls were improved with whole brain radiation therapy.
The oncologic benefit of postoperative stereotactic radiosurgery following surgical resection of brain metastasis as well established.
Postoperative SRS is associated with high rates of local control with minimum adverse effects, while avoiding a cognitive adverse effects of traditional whole brain radiation therapy.
Randomized clinical trials have shown their preoperative SRS has a comparable safety profile compared to postoperative SRT and results in short time to treatment completion.
Limited volume of metastases generally refers to 5-10 lesions or fewer.
Most patients with brain metastases have active systemic disease.
In a phase 2 trial (PASSPORT) trial for first and second line treatments of non-squamous cell lung cancer with previously treated brain metastases received bevacizumab with platinum or other single agent chemotherapy: There was no increase in CNS hemorrhage indicating that bevacizumab was a safe agent to be administered in patients with treated brain metastases (Socinski MA).
Of 359 patients with 1-3 brain metastases 199 underwent radiosurgery, and 160 underwent surgery: in the radiosurgery group 100 were observed and 99 underwent WBRT after surgery 79 patients were observed ad 81 received WBRT-WBRT reduced intracranial relapses and neurologic deaths but failed to improve the duration of functional independence and overall survival (Kocher M et al).
Whole brain radiation reduces CD4 counts from the normal range before radiation to less than 300 cells/mm3 in 47% of patients and less than 200 cells/mm3 in 26% of patients decreasing immune status systemically and within the brain (MulvennaPM).
An interim analysis of the CheckMate 920 phase IIIb/IV trial indicates that the combination of nivolumab and ipilimumab is safe and effective in advanced renal cell carcinoma (RCC) patients with brain metastases, with encouraging antitumor activity.
An interim analysis of the CheckMate 920 phase IIIb/IV trial indicates that the combination of nivolumab and ipilimumab is safe and effective in advanced renal cell carcinoma (RCC) patients with brain metastases, with encouraging antitumor activity.
The overall response rate was 29%, but no complete responses.
Targeted therapies are associated with improved survival in patients with brain metastasis and ERBB2 positive breast cancer, EGFR positive lung cancer, or BRAF positive melanoma.
Brainstem metastasis represent 4-7% of secondary intracranial malignancies, but they are not amenable to resection relating to high risk of morbidity and mortality: Sterotactic radiosurgery is an effective treatment for such lesions.
Brain metastases are associated with the thromboembolism: approximately 10% with lung cancer and lower with melanomas, renal cell cancer, breast cancer or colorectal cancer.
CSF ctDNA had a greater ability than plasma ctDNA to comprehensively represent the mutational landscape of brain metastases with CSF ctDNA detecting all BM mutations in 83.33% of patients, while plasma ctDNA was only 27.78%.
CSF ctDNA is superior to plasma ctDNA in accurately representing the profiling of single BM.
Plasma ctDNA could be an alternative liquid biopsy material to be applied in multiple brain metastatic NSCLC.
