 Increased susceptibility in patients above the age of 70 years and in smokers.
Increased susceptibility in patients above the age of 70 years and in smokers.
Starts gradually during treatment, but the development can occur up to 6 months after discontinuation of therapy.
In patients receiving a cumulative dose of <300 mg of bleomycin, the incidence of is 3 to 5%, while in patients treated with >500 mg, 20% develop interstitial pneumonitis.
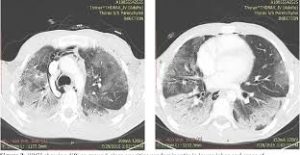
Diagnosis is one of exclusion since symptoms and radiographic imaging abnormalities are nonspecific.
Symptoms include shortness of breast, cough, tachycardia, cyanosis, and occasional fever.
Chest x-ray may show no changes or bilateral basilar infiltrates followed by diffuse interstitial and alveolar infiltrates.
Pneumothorax and pneumomediastinum may occur.
Endothelial damage due to cytokines and free radical formation with Fe 3+ complexes occur.
Age greater than 40 years, the presence of impaired renal function with glomerular filtration rate of <80 ml/min and a cumulative dose above 300 mg significantly associated with bleomycin pneumonitis.
Guidelines recommend baseline pulmonary function testing with measurement of the diffusing capacity of the lungs for carbon monoxide (DLCO).
The routine use of of granulocyte-colony-stimulating factor is not recommended.
Pulmonary function tests are the best tools for evaluating for pulmonary assessment.
The predictive value for DLCO for development of lung toxicity has been conflicting in studies.
Advisable to stop further bleomycin treatment when the diffusion capacity for carbon monoxide decreased more than 40-60%.
Symptomatic and fatal pulmonary toxic effects can be essentially eliminated by monitoring the diffusion capacity of the lung for common monoxide before each dose of Leo Mason and by stabbing therapy if the DLCO decreases.
High-dose corticosteroids, commonly used to treat bleomycin associated lung toxicity, although there are no randomized data supporting their use.
