 Automated insulin delivery systems are automated, or semi-automated, systems designed to assist people with diabetes, primarily type 1, by automatically adjusting insulin delivery to help them control their blood glucose levels.
Automated insulin delivery systems are automated, or semi-automated, systems designed to assist people with diabetes, primarily type 1, by automatically adjusting insulin delivery to help them control their blood glucose levels.
Artificial pancreatic technology mimics the secretion of these hormones into the bloodstream in response to the body’s changing blood glucose levels.
Maintaining balanced blood sugar levels is crucial to the function of the brain, liver, and kidneys.
For type 1 patients, the body cannot produce insulin itself.
Threshold suspend systems are the simplest form of insulin delivery automation.
They halt the constant flow of insulin from a pump when a connected glucose monitor reports a glucose level below a pre-set threshold.
It cannot remove insulin that has already been infused, so the overall efficacy of threshold suspend systems is limited due to the relatively slow pharmacokinetics of insulin delivered subcutaneously.
Predictive low glucose suspend (PLGS) systems use a mathematical model to extrapolate predicted future blood sugar levels based on recent past readings from a CGM.
This allows the system to halt insulin delivery as much as 30 minutes prior to a predicted hypoglycemic event, allowing addition time for the slow pharmacokinetics of insulin to reflect that delivery has been halted.
Hybrid closed loop (HCL) systems further expand on the capabilities of the predictive low glucose suspend systems, by adjusting basal insulin delivery rates both up and down in response to values from a continuous glucose monitor: the system is able to reduce the magnitude and duration both hyperglycemic and hypoglycemic events.
In a trial involving young children, with type1 diabetes, the glucose level was in the target range for greater percentage of time with a closed-loop system, than with standard care.
In addition to modulating basal insulin, advanced hybrid closed loop systems have the ability to deliver boluses of insulin to correct for elevated blood sugar.
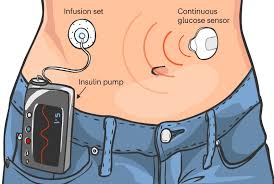
An automated insulin delivery system consists of three distinct components:
a continuous glucose monitor
a pump to deliver insulin,
and an algorithm that uses the data from the glucose monitor to send commands to the pump.
The algorithm ties the two devices together to close the loop and uses data regarding insulin delivery and glucose levels to adjust insulin dosing, typically on a five minute schedule.
Continuous glucose monitors (CGMs) are medical devices which extrapolate an estimate of the glucose concentration in a patient’s blood based on the level of glucose present in the subcutaneous interstitial fluid.
A thin, biocompatible sensor wire coated with a glucose-reactive enzyme is inserted into the skin, allowing the system to read the voltage generated, and based on it, estimate blood glucose.
CGM can take a new reading as often as every 60 seconds.
Most CGMs only take a reading every 5 minutes, providing current blood sugar level, but a record of past measurements; allowing computer systems to project past short-term trends into the future.
Continuous monitors use sensors placed just under the skin on the arm or abdomen to deliver blood sugar level data to receivers or smartphone apps as often as every few minutes.
The sensors can be used for up to fourteen days.
Hybrid closed loop system: Partially automates a patient’s basal insulin delivery.
It requires the inputting of basal rates, insulin sensitivity factors, carbohydrate to insulin ratios, the total daily dose of insulin, or subset of these metrics on initialization.
Insulin doses at meal time are determined by having the user enter the number of grams of carbohydrate in the meal, and effective therapy may be dependent on user initiated correction doses for hyperglycemia.
It is made up of a continuous glucose monitor, an insulin pump, and a glucose meter for calibration.
It automatically functions to modify the level of insulin delivery based on the detection of blood glucose levels by continuous monitor.
It does this by sending the blood glucose data through an algorithm that analyzes and makes the subsequent adjustments.
The system has two modes. Manual mode lets the user choose the rate at which basal insulin is delivered.
Auto mode regulates basal insulin levels from the CGM readings every five minutes.
Semi automated insulin delivery systems require individualized insulin regimens for the initiation of therapy and meal doses based on carbohydrate counting for a routine operation.
The bionic pancreas is initialized only on the basis of body weight, makes all those decisions and delivers insulin autonomously, and uses meal announcements without carbohydrate counting.
The device uses a closed-loop system to deliver both insulin and glucagon in response to sensed blood glucose levels.
The bionic pancreas does not use information about the patient’s previous insulin regimen, basal and bolus dose settings, but is initiaized only on the basis of body weight, and automates the determination and delivery of all insulin doses immediately after bodyweight data have been entered, with no warm-up period.
Meal announcements consist of an estimate of carbohydrate content as compared with the typical meal of that type.
The bionic pancreas continually adapts to the users insulin needs, end it is not possible for the user to determine or modify insulin doses.
Randomized trial involving adults and children with type one diabetes using a bionic pancreas was found to have a greater reduction than standard care in glycated hemoglobin levels. (Bionic pancreas research group).
Alternate controller enabled (ACE) insulin pumps allow users to integrate continuous glucose monitors, automated insulin dosing (AID) systems, and other diabetes management devices with the pump to create a personalized diabetes therapy system.
Closed-loop systems, unlike the continuous sensor alone, the closed-loop system requires no user input in response to reading from the monitor; the monitor and insulin pump system automatically delivers the correct amount of hormone calculated from the readings transmitted.
This system is what makes up the artificial pancreas device.
Open source automated insulin delivery and type one diabetes results in significantly higher percentage of time in the target glucose range than the use of sensor augmentef insulin pumps at 24 weeks.
The Bio-artificial pancreas is a bio-engineered tissue with encapsulated islet cells which deliver endocrine hormones in response to glucose.
Its membrane prevents antibodies of the patient from entering the device and damaging the encapsulated cells.
The surgically implanted membrane sheet is viable for years, and the cells that the device holds can be produced from stem cells rather than human donors, and may also be replaced over time using input and output connections without surgery.
These cells include insulin-producing beta cells, as well as alpha cells, which produce glucagon, mimicking the structure of the pancreas.
In a randomized 13 week control trial involving adults with insulin treated type two diabetes, automated insulin delivery was associated with the greater reduction in glycated hemoglobin levels than continuous glucose monitoring alone (21QP study group).
