Autoimmune diseases affect more than 5% of the world‘s population and involve virtually every organ system.
The major pathogenic mechanism involves IgG autoantibodies that recognize self antigens and produce pathologic effects by way of diverse pathways.
IgG is the most abundant antibody type in the bloodstream with a concentration of 10 g/L, second only to albumin with a concentration of 40 g/L.
The high concentration of IGG and albumin are due to their plasma half-life of 21 days which exceeds that of any other circulating protein.
Defects in immunoregulatory processes, especially V-region connectivity, are important in the development of autoimmune disease.
Autoimmune diseases are characterized by chronic inflammation, organ damage, and increased mortality.
As a result of autoreactive B cells that cause severe pathologic effects.
Affects 3% of the population.
An autoimmune disease is a condition arising from an abnormal immune response to a functioning body tissue.
Autoimmune diseases arise through aberrant reactions of the human adaptive or innate immune systems against the body’s own proteins.
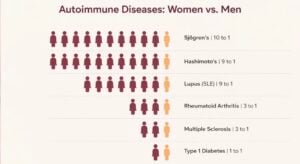
There may also be a hormonal component to autoantibody formation as many autoimmune conditions are much more prevalent in women of childbearing age.
There are at least 100 types of autoimmune diseases, that can affect any body part of the body.
Overall cumulative US prevalence for the autoimmune diseases at 5.0%, with 3.0% for males and 7.1% for females.
Common symptoms include low grade fever and fatigue.
Symptoms come and go.
The usual onset of symptoms is during adulthood.
Symptoms of early autoimmune disease are often the same as common illnesses, including: fatigue, fever, malaise, joint pain, and rash.
Autoimmune diseases: alopecia areata, celiac disease, diabetes mellitus type 1, Graves’ disease, inflammatory bowel disease, multiple sclerosis, psoriasis, rheumatoid arthritis, systemic lupus erythematosus, and others.
The cause is generally unknown:
some autoimmune diseases such as lupus run in families, and certain cases may be triggered by infections or other environmental factors.
It is suggested up to seventy percent of autoimmune disease are due to environmental factors, including: chemicals, infection, diet, and gut dysbiosis.
Treatment depends on the type and severity of the autoimmune condition: Nonsteroidal anti-inflammatory drugs (NSAIDs), immunosuppressants, and Intravenous immunoglobulin may also occasionally be used.
Treatment usually improves symptoms, they typically does not cure the disease.
Women are more commonly affected than men.
AD usually starts during adulthood.
Autoimmune diseases present similar symptoms: appearance and severity of these signs and symptoms depends on the location and type of autoimmune response that occurs.
Patients may have more than one autoimmune disease simultaneously, and display symptoms of multiple diseases.
Treatment targeting B cells in auto immune disease, attempts to either deplete B cells or inhibit their activation.
Cd-19 CAR-T cell transfer is a safe and efficacious treatment in auto immune disorders.
The disease and signs and symptoms presentation can be influenced by various age, hormones, and environmental factors.
Common symptoms of AD:
Fatigue
Low grade fever
Malaise
Muscle aches and joint pain
Rash
Signs and symptoms can fluctuate.
When signs and symptoms reappear, it is known as a flare-up.
Such signs and symptoms may aid in diagnosis by supporting the results from biologic markers of autoimmune diseases.
Tissues commonly impacted by autoimmune diseases: blood vessels, connective tissues, joints and muscles, red blood cells, skin, and endocrine glands, like thyroid or pancreas glands.
Associated conditions include vasculitis and anemia.
Symptoms will vary from person to person, vary over time, vary with organ involvement, and they may taper off or flare unexpectedly.
Patients may have more than one autoantibody, and thus have more than one autoimmune disorder, and/or have an autoimmune disorder without a detectable level of an autoantibody, complicating making a diagnosis.
Frequency 24 million / 7% in the US.
Mechanisms whereby autoantibodies result in clinical disease is mostly unexplained.
Between 20 and 50 million people in the US have an autoimmune disease.
Organ specific autoanti-bodies are common among the elderly.
Old age is complicated by an increase in autoimmune disease frequency.
The pathological effects that characterize them as an autoimmune disease: damage to or destruction of tissues where there is an abnormal immune response, altered organ growth, and altered organ function depending on the location of the disease.
Autoimmune disease can be organ specific and are restricted to affecting certain tissues, while others are systemic diseases that impact many tissues throughout the body.
Signs and symptoms may vary with an individuals disease.
An autoimmune disease increases the risk or likelihood of developing certain cancers.
Cancer risk of autoimmune diseases is increased by chronic inflammation which has been linked to cancer.
The autoimmune diseases most commonly linked to cancer including celiac disease, inflammatory bowel disease, multiple sclerosis, rheumatoid arthritis, and systemic lupus erythematosus.
Celiac disease presents the strongest associations to gastrointestinal and lymphoproliferative cancers.
In celiac disease, the autoimmune reaction is caused by the body’s loss of immune tolerance to ingested gluten, found primarily in wheat, barley, and rye:
increased risk of gastrointestinal cancers, as the gastrointestinal tract includes the esophagus, stomach, small intestine, large intestine, rectum, and anus, all areas that the ingested gluten would traverse in digestion.
The incidence of gastrointestinal cancer can be reduced or eliminated if a patient removes gluten from their diet.
Inflammatory bowel disease is associated with cancers of the gastrointestinal tract and some lymphoproliferative cancers due to loss of immune tolerance.
Inflammatory bowel disease individuals lose immune tolerance for normal bacteria present in the gut microbiome, and the immune system attacks the bacteria and induces chronic inflammation, which has been linked to increased cancer risk.
Multiple sclerosis is associated with decreased risk of cancer overall but an increased risk of central nervous system cancer, primarily in the brain.
Multiple sclerosis is a neurodegenerative disease in which T-cells attack the important myelin sheath in brain neurons, which reduces the nervous system function, creating inflammation and subsequent cancer of the brain.
Rheumatoid arthritis has significant associations with focal cancers all throughout the body and lymphoproliferative cancers.
In rheumatoid arthritis the chronic inflammation and overactivation of the immune system creates an environment that favors malignant transformation of other cells.
Systemic lupus erythematosus is associated with focal cancers throughout the body and lymphoproliferative cancers.
Systemic lupus erythematosus is characterized by a widespread loss of immune tolerance.
Conversely, systemic lupus erythematosus is correlated with a decrease in some cancers.
There are more than 100 autoimmune diseases.
The exact genes responsible for causing each autoimmune disease have not been completely elucidated.
With Type 1 diabetes pancreatic ?-cells are targeted and destroyed by the immune system, as a result of neo-natal mutations to the insulin gene (INS) which is responsible for mediating the production of the insulin in the pancreas.
The insulin gene is located on the short arm of chromosome 11.
In addition to chromosome 11, a genetic determinant of type 1 diabetes is a locus called the major histocompatibility complex (MHC) located on chromosome 6p21.
Several genes are thought to play a role in causing rheumatoid arthritis.
B1 genes in the human genome contain an increased concentration of HLA-DRB1 alleles that are most commonly seen in RA patients.
RA can vary in severity as a consequence of polymorphisms within the genome.
Environmental factors have a direct role in development, or are catalysts for many autoimmune diseases.
Chemicals in the environment, in the form of drugs, hydrazines, hair dyes, trichloroethylene, tartrazines, hazardous wastes, and industrial emissions may be associated with ADs..
UV radiation exposure a possible cause of development of the autoimmune disease dermatomyositis.
Pesticides may play a role in the development of rheumatoid arthritis.
Infectious agents that activate T cells may be associated with autoimmune diseases, suggested in Guillain-Barre syndrome and rheumatic fever.
Staphylococcal and streptococcal superantigens have been characterized in autoimmune diseases, with similar proteins to body tissues.
The human immune system typically produces both T cells and B cells that are capable of being reactive with self-antigens.
In ((immune tolerance)) self-reactive cells are usually either killed prior to becoming active within the immune system, placed into a state of anergy, or removed from their role within the immune system by regulatory cells.
Regulatory T cells are defective in many autoimmune diseases.
With AD any one of these mechanisms can fail, with residual self-reactive cells.
The prevention of self-reactive T cells occurs by negative selection process within the thymus as the T cell is developing into a mature immune cell.
Some infections, such as Campylobacter jejuni, have antigens that are similar to our own self-molecules, resulting in the production of antibodies that also react to a lesser degree with gangliosides of myelin sheath surrounding peripheral nerves’ axons.
Autoimmunity refers to the presence of self-reactive immune response: auto-antibodies, self-reactive T cells, with or without damage or pathology resulting from it.
Autoimmunity may be restricted to certain organs, such as in in autoimmune thyroiditis. or involve tissues in different places, such as Goodpasture’s disease, affecting the basement membrane in both the lung and the kidney.
No single test can identify an autoimmune disease.
Antinuclear antibody testing isused to identify abnormal proteins, antinuclear antibodies, produced when the body attacks its own tissues.
The antinuclear antibody test is most useful for diagnosing systemic lupus erythematosus, having a 95% positive test rate.
Complete blood count may be associated with low white blood cell count.
If complement levels are low, this may be an indication of autoimmune disease.
C reactive protein generally increases with inflammation, and may be high in autoimmune disease.
Erythrocyte sedimentation rate measures the rate at which a patient’s blood cells descend in a test tube, with a more rapid descent a common finding in autoimmune disease.
Treatment varies with the type and severity of the condition.
The majority of the autoimmune diseases are chronic with no known cure.
Most autoimmune disease symptoms can be alleviated and controlled with treatment.
Goals of treatment: lessen symptoms, manipulate thebautoimmune response, and preserve the ability of the patient to combat diseases that they may encounter.
Primary treatment options may include immunosuppressant drugs to weaken the overall immune response:
Non-steroidal anti-inflammatory drugs (NSAIDs) to reduce inflammation
Glucocorticoids to reduce inflammation
Disease-modifying anti-rheumatic drugs (DMARDs) to decrease the damaging tissue and organ effects of the inflammatory autoimmune response
Physical therapy if the disease impacts bones, joints, or muscles.
Monoclonal antibodies that can be used to block pro-inflammatory cytokines.
Antigen-specific immunotherapy which allows immune cells to specifically target the abnormal cells that cause autoimmune disease.
CAR T therapy being explored for autoimmune disease.
Many people have more than one autoimmune disease: 4.5% overall, with 2.7% for males and 6.4% for females.
Autoantibodies that have been initialized, have the capacity to maintain their own production.
Individuals with autoimmunity have alterations in their glycosylation profile such that a proinflammatory immune response is favored.
The hygiene hypothesis of autoimmune disease: high levels of cleanliness expose children to fewer antigens, causing their immune systems to become overactive and more likely to misidentify own tissues as foreign, resulting in autoimmune or allergic conditions such as asthma.
There is a well-established association between autoimmune rheumatic diseases and atherosclerotic cardiovascular disease.
The link between autoimmune disease and cardiovascular disease is related to an increased expression of pro inflammatory cytokines, pro thrombotic mediators, up regulation of oxidative stress, and abnormal vascular tone.
Diagnostic tests:
blood tests to detect inflammation, autoantibodies, and organ involvement
x-rays and other imaging scans to detect changes in bones, joints, and organs
biopsies to look for pathologic changes in tissue specimens

One reply on “Autoimmune disease”
I appreciate, result in I found just what I was taking a look for. You have ended my 4 day lengthy hunt! God Bless you man. Have a nice day. Bye